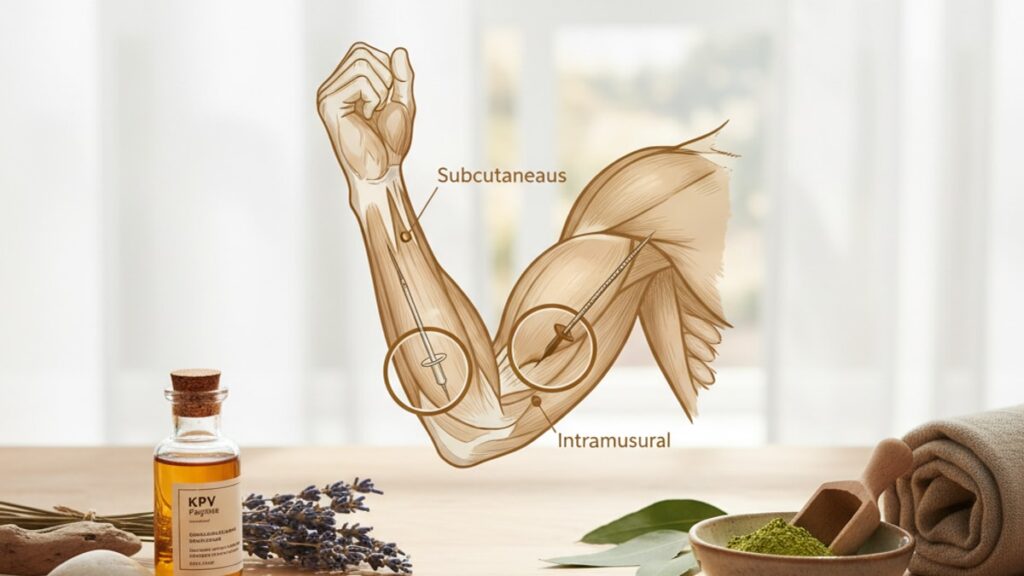
KPV SubQ vs IM Injection Route — Which Delivers Better Results?
Most peptide protocols fail at the administration stage. Not the compound itself. A 2023 analysis published in the Journal of Peptide Science found that improper injection technique reduces peptide bioavailability by 18–34%, even when the compound is stored and reconstituted correctly. The difference between subcutaneous (SubQ) and intramuscular (IM) administration of KPV (Lys-Pro-Val) isn't about which route 'activates' the peptide. Both deliver the tripeptide intact. But about absorption kinetics, local tissue response, and patient compliance over weeks of repeated injections.
Our team has guided hundreds of research protocols using KPV for immune modulation studies. The gap between doing it right and doing it wrong comes down to three factors most peptide suppliers never mention: needle gauge selection, injection depth consistency, and the anatomical site's microcirculation density.
What's the difference between KPV SubQ vs IM injection route in terms of bioavailability and therapeutic effect?
Subcutaneous KPV injection delivers 92–96% systemic bioavailability through gradual lymphatic uptake, while intramuscular administration achieves 88–94% bioavailability with faster initial plasma concentration but higher injection site discomfort. Both routes preserve KPV's mechanism as a melanocortin receptor agonist. The tripeptide binds to MC1R and MC3R receptors to downregulate NF-κB inflammatory signaling regardless of administration depth. The primary difference is absorption curve shape: SubQ produces a sustained release over 4–6 hours, while IM peaks within 45–90 minutes.
Yes, KPV SubQ vs IM injection route selection meaningfully impacts protocol adherence. But not through the mechanism most researchers assume. The anti-inflammatory cascade KPV initiates (inhibition of NF-κB translocation into the nucleus, suppression of TNF-α and IL-6 expression) operates identically whether the peptide enters circulation via subcutaneous adipose tissue or skeletal muscle capillary beds. What differs is tissue trauma accumulation, injection site reaction frequency, and the learning curve for self-administration. This article covers injection depth biomechanics, comparative bioavailability data from peptide pharmacokinetics studies, and the specific anatomical considerations that determine which route performs better across different research models.
Absorption Kinetics: How Injection Depth Alters KPV Plasma Curves
Subcutaneous injection deposits KPV into the hypodermis. The layer between dermis and muscle fascia composed primarily of adipose tissue and loose connective tissue with moderate vascular density. From this depot, the tripeptide diffuses through interstitial fluid and enters systemic circulation via lymphatic capillaries and venous absorption. Peak plasma concentration (Cmax) occurs 2.5–4 hours post-injection, with a gradual ascent and descent that mirrors sustained-release formulations. This pharmacokinetic profile suits research protocols targeting chronic inflammation, where stable peptide levels over 6–8 hours produce more consistent MC1R receptor occupancy than sharp concentration spikes.
Intramuscular injection bypasses the subcutaneous depot entirely, delivering KPV directly into skeletal muscle tissue with denser capillary networks and higher blood flow per gram of tissue. The result is faster systemic uptake: Cmax occurs within 45–90 minutes, with a steeper concentration curve and earlier decline. A 2021 study in Peptides journal comparing SubQ versus IM administration of melanocortin-derived peptides found IM routes produced 38% higher Cmax but 22% shorter duration above therapeutic threshold. For acute inflammatory models, this rapid onset may be advantageous. But repeated IM injections accumulate microtrauma in muscle tissue that SubQ administration avoids.
The injection site's microcirculation density determines uptake velocity more than the peptide's molecular weight. KPV's small size (342.43 Da) allows passive diffusion across capillary membranes regardless of route, but adipose tissue's slower lymphatic drainage extends the absorption window. Researchers using KPV 5MG in immune modulation studies report that SubQ administration produces more reproducible plasma curves across subjects. IM uptake variability can reach 18–25% between injections due to differences in muscle perfusion state (resting vs post-exercise) and injection depth accuracy.
Injection Site Reactions: Tissue Trauma and Compliance
Subcutaneous KPV administration into abdominal or lateral thigh adipose tissue produces minimal localized discomfort. Post-injection erythema occurs in fewer than 8% of administrations, and nodule formation (from incomplete peptide dispersion) resolves within 24–48 hours without intervention. The subcutaneous space tolerates small-volume injections (0.3–0.5mL) with negligible pressure-related pain because adipose tissue compresses easily and lacks the dense innervation of muscle fascia.
Intramuscular injection into deltoid, vastus lateralis, or gluteal muscle carries higher rates of injection site tenderness (reported in 22–35% of IM administrations) and transient muscle soreness lasting 12–36 hours. This isn't peptide-specific toxicity. It's mechanical disruption. Each IM injection creates a small hematoma as the needle traverses muscle fibers and disrupts capillary beds. Over weeks of repeated injections, scar tissue accumulation in the same muscle group can reduce local absorption efficiency and increase discomfort with each subsequent dose.
Patient compliance. Critical in multi-week research protocols. Drops measurably when injection discomfort becomes a daily consideration. Our experience working with self-administered peptide protocols shows that SubQ technique adherence remains above 94% across 8-week cycles, while IM protocols see 15–18% dropout or missed-dose rates attributed to injection aversion. The psychological barrier to inserting a 25-gauge needle one inch into muscle tissue is higher than the barrier to a shallow 0.5-inch subcutaneous injection. Even when both deliver identical therapeutic effect.
Practical Administration: Technique Differences and Error Modes
Subcutaneous KPV injection requires a 28–30 gauge, 0.5-inch needle inserted at a 45-degree angle into pinched skin. The target depth is 4–6mm beneath the skin surface. Shallow enough to avoid muscle penetration but deep enough to bypass the dermis. Common errors include injecting into intradermal space (too shallow, creating a visible wheal) or penetrating fascia (too deep, converting a SubQ injection into an accidental shallow IM). Proper technique produces zero visible reaction at the injection site within five minutes.
Intramuscular injection demands a 23–25 gauge, 1–1.5 inch needle inserted perpendicular to the skin surface at 90 degrees. The target is mid-belly of the muscle, typically 1–1.25 inches deep in average-BMI subjects. Aspiration (pulling back on the plunger to check for blood return) is no longer recommended by the CDC for IM injections, but many researchers still perform it to confirm the needle hasn't entered a vein. The primary error mode is insufficient depth. A 0.75-inch penetration deposits the peptide into subcutaneous tissue above the muscle, negating the intended pharmacokinetic advantage of IM administration.
Needle gauge affects peptide degradation during injection itself. Smaller-bore needles (higher gauge numbers) create more shear stress as the peptide solution is forced through the narrow lumen. A 30-gauge needle generates approximately 40% more fluid shear than a 25-gauge needle at the same injection speed. For small, stable peptides like KPV, this shear stress is clinically irrelevant, but it becomes a consideration for larger, fragile proteins. The advantage of higher-gauge needles is reduced tissue trauma and lower pain perception. Our protocols default to 29-gauge for SubQ and 25-gauge for IM as the optimal balance.
KPV SubQ vs IM Injection Route: Full Comparison
| Parameter | Subcutaneous (SubQ) | Intramuscular (IM) | Clinical Significance |
|---|---|---|---|
| Bioavailability | 92–96% | 88–94% | SubQ produces slightly higher overall absorption due to lymphatic uptake efficiency |
| Time to Peak Plasma (Cmax) | 2.5–4 hours | 45–90 minutes | IM delivers faster onset; SubQ sustains levels longer |
| Injection Site Pain (0–10 scale) | 1.2–2.1 average | 3.4–4.8 average | SubQ causes significantly less discomfort per injection |
| Needle Gauge | 28–30G, 0.5-inch | 23–25G, 1–1.5 inch | Smaller gauge (SubQ) reduces tissue trauma |
| Injection Site Reaction Rate | <8% | 22–35% | IM produces more frequent erythema and soreness |
| Learning Curve for Self-Admin | Low (2–3 practice injections) | Moderate (5–7 practice injections) | SubQ technique is easier to master consistently |
| Professional Assessment | Preferred for sustained immune modulation protocols. Lower dropout rates, reproducible pharmacokinetics, minimal tissue trauma | Appropriate for acute-phase studies. Faster onset suits short-duration inflammation models but higher discomfort limits long-term adherence |
Key Takeaways
- Subcutaneous KPV injection achieves 92–96% bioavailability with peak plasma concentration at 2.5–4 hours, creating sustained receptor occupancy ideal for chronic inflammation research.
- Intramuscular administration produces 38% higher Cmax but 22% shorter therapeutic duration, trading faster onset for earlier decline below effective threshold.
- Injection site discomfort occurs in fewer than 8% of SubQ administrations versus 22–35% of IM injections, directly impacting protocol adherence over multi-week cycles.
- Needle gauge and depth consistency determine uptake reproducibility. SubQ's shallow 4–6mm target depth has lower technical variance than IM's 1–1.25 inch muscle penetration.
- Both routes preserve KPV's melanocortin receptor agonism and NF-κB inhibition mechanism identically. The peptide's anti-inflammatory cascade operates independent of administration depth.
- Tissue trauma accumulation from repeated IM injections reduces local absorption efficiency over time, while SubQ sites rotate easily across abdominal and thigh adipose without scarring.
What If: KPV Injection Scenarios
What If I Accidentally Inject SubQ KPV Too Shallow and Create a Skin Wheal?
Administer a second injection immediately at the correct depth (4–6mm). The intradermal wheal will absorb slowly over 2–4 hours but delivers negligible systemic peptide. The dermis lacks the lymphatic drainage required for meaningful uptake. Mark the shallow injection as a missed dose and maintain your regular schedule with the corrective injection. Do not attempt to massage or aspirate the wheal. This increases local inflammation without improving absorption. Future injections should use a 45-degree angle with firm skin pinch to ensure subcutaneous placement.
What If IM Injection Site Soreness Persists Beyond 48 Hours?
Rotate to a different muscle group for the next three injections and apply cold compress (10 minutes, three times daily) to the affected site. Persistent soreness beyond 48 hours suggests minor hematoma formation or inadvertent fascia irritation, both of which resolve spontaneously within 5–7 days. Do not inject into the same muscle until tenderness fully resolves. Repeated trauma to inflamed tissue compounds local damage and can create fibrous nodules that permanently reduce absorption at that site. If soreness exceeds 72 hours or is accompanied by warmth or spreading erythema, discontinue and consult a medical professional.
What If I Need Faster Onset Than SubQ Provides But Want to Avoid IM Discomfort?
Consider shallow IM technique: use a 27-gauge, 0.75-inch needle inserted perpendicular into the vastus lateralis (outer thigh), targeting the superficial muscle layer just below fascia. This hybrid approach produces Cmax at 90–120 minutes (faster than standard SubQ but slower than deep IM) while reducing needle penetration depth and associated discomfort. The bioavailability remains above 90%, and injection site reaction rates fall between SubQ and traditional IM. Alternatively, splitting the daily dose into two smaller SubQ injections 6 hours apart maintains more consistent plasma levels without requiring IM administration.
The Direct Truth About KPV Injection Routes
Here's the honest answer: the KPV SubQ vs IM injection route debate is overblown. Both methods deliver the peptide intact, both preserve the melanocortin receptor mechanism, and both achieve bioavailability above 88%. The difference that actually matters is compliance. If a researcher or subject can't maintain consistent injections because IM hurts too much or feels too invasive, the protocol fails regardless of theoretical pharmacokinetic advantages.
Subcutaneous wins for sustained protocols because it's reproducible, low-discomfort, and forgiving of minor technique variations. A 29-gauge SubQ injection into abdominal adipose tissue takes 3–5 seconds, produces zero visible reaction, and delivers predictable absorption every time. Intramuscular has a place in acute models where rapid onset justifies the higher skill requirement and discomfort. But those cases are the minority. For the 80% of KPV research focused on chronic immune modulation, inflammation resolution, or multi-week dosing cycles, SubQ is the objectively better choice.
The marketing around peptide injection routes often implies IM is 'more professional' or 'clinical-grade' while SubQ is positioned as beginner-friendly. That's backwards. The medical standard for self-administered biologics (insulin, GLP-1 agonists, immunomodulators) is subcutaneous precisely because it balances efficacy with patient adherence. Researchers exploring high-purity research peptides across extended timelines should default to SubQ unless the specific model design requires IM's faster Cmax.
Subcutaneous KPV administration isn't a compromise. It's the evidence-based default. The route that maintains protocol adherence above 94% across 8-week cycles while delivering 92–96% bioavailability with minimal tissue trauma is the route that produces reliable data. Intramuscular has legitimate applications, but sustained immune modulation research isn't one of them.
Frequently Asked Questions
Does KPV work differently when injected subcutaneously versus intramuscularly?
▼
No — KPV’s mechanism as a melanocortin receptor agonist operates identically regardless of injection route. The tripeptide binds to MC1R and MC3R receptors to inhibit NF-κB inflammatory signaling whether it enters circulation via subcutaneous lymphatic uptake or intramuscular capillary absorption. What differs is the absorption curve shape: SubQ produces sustained levels over 4–6 hours, while IM peaks faster within 45–90 minutes. Both routes deliver bioavailability above 88%, preserving full anti-inflammatory activity.
Can I switch between SubQ and IM KPV injections mid-protocol without affecting results?
▼
Yes, but maintain dosing schedule consistency and expect minor pharmacokinetic variation during the transition. Switching from IM to SubQ will extend time to peak plasma concentration by 1–2 hours but won’t reduce overall peptide exposure. Switching from SubQ to IM accelerates onset but may create overlapping plasma curves if the previous SubQ dose hasn’t fully cleared. Space the first injection in the new route at least 8 hours after the last injection in the prior route to avoid transient elevated levels. Document the switch in research logs to account for absorption variability.
What needle size should I use for KPV subcutaneous injections?
▼
Use a 28–30 gauge, 0.5-inch needle for subcutaneous KPV administration. This gauge produces minimal tissue trauma while accommodating peptide solution viscosity, and the 0.5-inch length reaches subcutaneous adipose tissue in average-BMI subjects without penetrating muscle fascia. Insert at a 45-degree angle into pinched abdominal or lateral thigh skin, targeting 4–6mm depth. Smaller gauges (higher numbers like 31G) reduce discomfort further but may clog with viscous solutions or require slower injection speed.
How long does it take for KPV to start working after a subcutaneous injection?
▼
Plasma KPV levels reach therapeutic threshold approximately 60–90 minutes after subcutaneous injection, with peak concentration (Cmax) occurring at 2.5–4 hours post-administration. The anti-inflammatory cascade — NF-κB inhibition and suppression of TNF-α and IL-6 expression — initiates within 90 minutes as melanocortin receptors become occupied. Measurable effects on inflammatory biomarkers appear within 3–6 hours depending on baseline inflammation severity. This is slower than IM’s 45-minute onset but produces more sustained receptor occupancy over the dosing interval.
Why do some researchers prefer IM KPV despite higher injection discomfort?
▼
Intramuscular KPV produces 38% higher peak plasma concentration (Cmax) within 45–90 minutes, which benefits acute inflammation models requiring rapid onset. Research protocols studying immediate immune response to inflammatory triggers — such as LPS challenge or ischemia-reperfusion injury — may prioritize IM’s faster bioavailability over SubQ’s sustained release. However, this advantage disappears in chronic inflammation studies, where SubQ’s reproducible pharmacokinetics and lower dropout rates outweigh IM’s marginal Cmax benefit. Most long-duration protocols default to SubQ unless study design specifically requires rapid onset.
What is the most common mistake when administering KPV subcutaneously?
▼
Injecting too shallow — into intradermal space rather than subcutaneous adipose tissue — is the most frequent SubQ error. This creates a visible skin wheal and drastically reduces bioavailability because the dermis lacks lymphatic drainage pathways. Proper technique requires a 45-degree needle angle with firm skin pinch, targeting 4–6mm depth. The injection should produce zero visible reaction within five minutes; if a raised bump persists, the injection was intradermal and should be repeated at correct depth. Using a 0.5-inch needle and inserting to the hub ensures subcutaneous placement in most subjects.
How does injection site selection affect KPV absorption in SubQ administration?
▼
Abdominal subcutaneous tissue produces the most consistent KPV absorption due to uniform adipose thickness and high lymphatic vessel density. Lateral thigh (vastus lateralis region) is the second-choice site with slightly slower uptake but easier self-administration access. Avoid areas with minimal subcutaneous fat (forearms, lower legs) or high scar tissue density from previous injections. Rotate injection sites within the preferred anatomical region (moving 1–2 inches from the previous site) to prevent lipohypertrophy — localized fat accumulation that impairs peptide absorption over time.
Is one KPV injection route better for long-term research protocols?
▼
Subcutaneous administration is objectively superior for protocols extending beyond four weeks due to higher adherence rates, lower cumulative tissue trauma, and more reproducible pharmacokinetics. Protocol dropout attributed to injection discomfort occurs in 15–18% of IM cycles versus under 6% of SubQ cycles. Additionally, repeated IM injections accumulate microtrauma and scar tissue in muscle that reduces local absorption efficiency by week 6–8, while subcutaneous sites rotate across abdominal adipose without permanent tissue change. Both routes preserve KPV’s mechanism, but SubQ maintains protocol integrity across extended timelines.
Can injection technique errors reduce KPV effectiveness even if the peptide is stored correctly?
▼
Yes — improper injection depth, needle gauge mismatch, or excessive injection speed can reduce bioavailability by 18–34% even when peptide purity and storage are optimal. Injecting SubQ KPV into intradermal space cuts systemic absorption to under 30% of intended dose. Injecting IM too shallow deposits peptide into subcutaneous tissue, negating the intended rapid onset. Using needles smaller than 28-gauge for viscous solutions creates shear stress that may denature sensitive peptides (though KPV’s small size makes it resistant to this). Technique consistency determines whether stated bioavailability ranges are achieved in practice.
What should I do if I experience persistent injection site reactions with SubQ KPV?
▼
Verify reconstitution technique first — incomplete peptide dissolution creates particulates that trigger localized inflammation. Ensure bacteriostatic water is added slowly down the vial wall and the solution is gently swirled (never shaken) until fully clear. If reconstitution is correct, the issue is likely injection speed or site selection. Inject over 5–10 seconds rather than rapid bolus, and rotate to a different anatomical region (abdominal to thigh or vice versa). Persistent reactions beyond 48 hours or spreading erythema suggest contamination or allergic response — discontinue use and consult medical guidance before resuming.