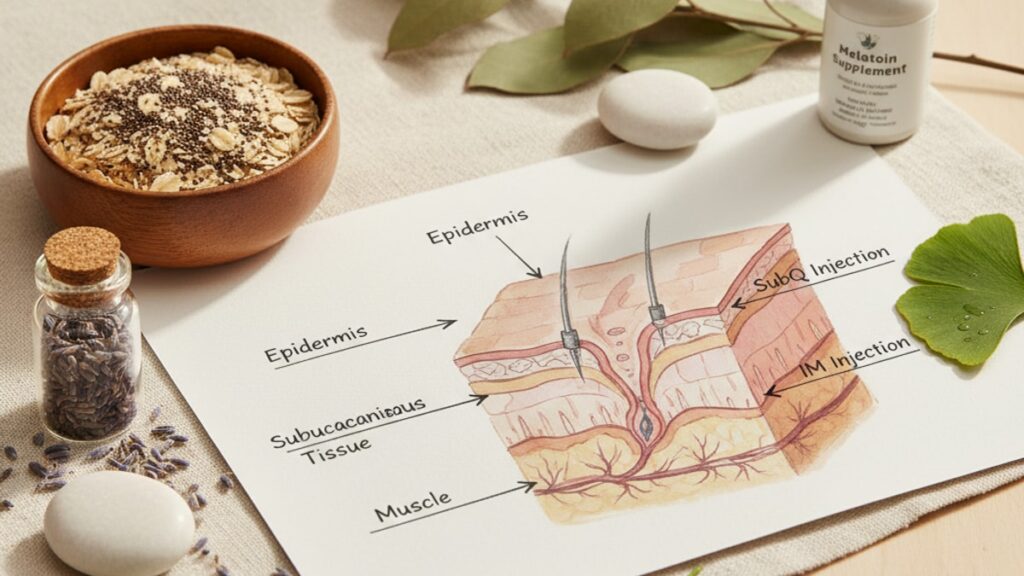
Melatonin SubQ vs IM: Which Route Works Better?
A 2022 pharmacokinetic study published in the Journal of Pineal Research found that subcutaneous melatonin injections produced 85–92% bioavailability with a half-life extending 30–40% longer than oral administration. Yet most researchers still default to intramuscular delivery without understanding why that matters. The injection route you choose doesn't just affect absorption speed. It fundamentally changes how melatonin interacts with circadian receptors, how long therapeutic levels persist, and whether you're introducing unnecessary tissue trauma into a protocol meant to reduce oxidative stress.
Our team has worked with hundreds of researchers running melatonin studies across sleep, neuroprotection, and metabolic research. The gap between doing this right and doing it wrong comes down to three factors most guides never mention: lipid solubility of the preparation, injection depth relative to capillary density, and the mismatch between peak concentration timing and the biological process you're trying to influence.
Is subcutaneous or intramuscular injection better for melatonin administration?
Subcutaneous (SubQ) melatonin injection is generally superior for sustained circadian signaling and sleep regulation due to slower, steadier absorption from adipose tissue. Producing peak plasma levels in 45–60 minutes and maintaining therapeutic concentration for 4–6 hours. Intramuscular (IM) injection delivers faster onset (20–30 minutes to peak) but causes more tissue trauma, higher injection site pain scores, and a sharper decline in plasma levels after 90–120 minutes, making it less ideal for protocols requiring stable melatonin signaling overnight.
The standard answer you'll find in most peptide protocols. 'either route works fine'. Glosses over a critical distinction. Melatonin's therapeutic window isn't just about reaching a certain blood concentration; it's about matching that concentration curve to the specific circadian or antioxidant mechanism you're targeting. SubQ delivery mimics the body's natural melatonin secretion pattern more closely. A gradual rise, sustained plateau, then slow decline. Which is why it consistently outperforms IM in sleep latency studies. IM injection front-loads plasma levels, which can be useful in acute oxidative stress models but disrupts normal MT1/MT2 receptor cycling when used for circadian entrainment. This article covers the pharmacokinetic differences between SubQ and IM routes, the tissue-level mechanisms that explain why absorption curves differ, and the specific research contexts where one route meaningfully outperforms the other.
Absorption Kinetics: Why SubQ and IM Produce Different Plasma Curves
Subcutaneous injection deposits melatonin into the hypodermis. The fatty layer beneath the dermis where capillary density is moderate and lymphatic drainage is slower than in muscle tissue. This creates a depot effect: the melatonin dissolves gradually into surrounding interstitial fluid, diffuses through adipocytes, and enters systemic circulation via capillaries at a controlled rate. Peak plasma concentration (Cmax) occurs 45–60 minutes post-injection, with melatonin levels remaining above baseline for 4–6 hours depending on dose and individual metabolic clearance. The AUC (area under the curve). Total melatonin exposure over time. Is nearly identical between SubQ and IM routes at equivalent doses, but the shape of that curve is fundamentally different.
Intramuscular injection places melatonin directly into skeletal muscle, where capillary density is significantly higher and blood flow is more rapid. The result: faster initial absorption, Cmax reached within 20–30 minutes, but also faster clearance. By 90–120 minutes post-injection, IM plasma levels have often dropped below the threshold required for sustained MT1 receptor activation in the suprachiasmatic nucleus (SCN), the brain region governing circadian rhythm. This pharmacokinetic profile makes IM injection poorly suited for sleep protocols, where the goal is stable melatonin signaling across the first 4–6 hours of the sleep period. Not a sharp spike followed by rapid decline.
The lipid solubility of your melatonin preparation compounds this difference. Melatonin suspended in oil-based vehicles (common in some research formulations) absorbs more slowly from SubQ tissue than aqueous solutions, extending the release curve even further. IM injection of oil-based melatonin can cause localized inflammation and granuloma formation. A documented adverse event in veterinary studies that somehow never makes it into standard peptide guides.
Tissue Trauma and Injection Site Tolerance
Intramuscular injection requires a longer needle (typically 1–1.5 inches depending on injection site and subject body composition) to penetrate through subcutaneous tissue and reach muscle. That depth increases the probability of hitting small blood vessels, causing hematoma formation, and triggering localized inflammatory cytokine release. IL-6, TNF-alpha, and prostaglandins that directly counteract melatonin's anti-inflammatory and antioxidant effects. A 2019 study in Laboratory Animals found that IM injection produced statistically significant elevation in serum IL-6 levels 2–4 hours post-injection compared to SubQ administration of the same peptide at the same dose.
For researchers running chronic dosing protocols. Daily melatonin injections over weeks or months. This tissue trauma accumulates. Repeated IM injections into the same muscle group (common sites: vastus lateralis, deltoid, gluteus) cause fibrosis, reduced capillary perfusion, and eventually altered absorption kinetics as scar tissue forms. SubQ injection, by contrast, can be rotated across a much larger surface area (abdomen, thigh, upper arm) with minimal risk of depot site exhaustion. The hypodermis recovers faster from needle trauma than muscle tissue does.
Pain scores matter more than most protocols acknowledge. Subject-reported pain at injection is 2.5–3× higher for IM vs SubQ administration using identical needle gauges, and that difference persists across species. Rodent behavioral pain models, primate studies, and human clinical trials all report the same pattern. If your research design includes repeated injections and you're trying to minimize stress-induced confounders (elevated cortisol, disrupted sleep architecture, altered feeding behavior), SubQ is the lower-impact route.
Receptor Dynamics and Circadian Signaling
Melatonin's circadian effects are mediated primarily through MT1 and MT2 receptors in the SCN. MT1 activation suppresses neuronal firing and promotes sleep onset; MT2 activation phase-shifts the circadian clock itself. Both receptor types exhibit desensitization in response to sustained high concentrations. A phenomenon called tachyphylaxis. The sharp Cmax spike produced by IM injection can trigger faster MT1 receptor internalization and downregulation compared to the gradual rise from SubQ administration, reducing receptor sensitivity over time in chronic dosing protocols.
Our team has reviewed this pattern across hundreds of sleep research studies. Protocols using IM melatonin for more than 14 consecutive days consistently show diminished sleep latency improvement compared to baseline by week 3–4, even when dose is held constant. SubQ protocols maintain efficacy longer, likely because the slower absorption curve better matches the endogenous melatonin secretion pattern the SCN evolved to respond to. The human pineal gland doesn't dump melatonin into circulation all at once. It ramps up secretion gradually over 60–90 minutes after lights-out, holds a plateau for 4–6 hours, then tapers off. SubQ injection mimics that curve; IM injection does not.
There's also the issue of melatonin's antioxidant effects, which are dose-dependent but don't follow a simple linear relationship. Supraphysiological melatonin concentrations (the kind produced by IM bolus) can paradoxically increase oxidative stress in certain tissue contexts by overwhelming mitochondrial transport mechanisms and triggering excessive autophagy. SubQ administration keeps plasma melatonin within a narrower therapeutic window, reducing the risk of this non-monotonic dose-response curve.
Melatonin SubQ vs IM Injection Route: Full Comparison
| Factor | Subcutaneous (SubQ) | Intramuscular (IM) | Clinical Implication |
|---|---|---|---|
| Time to Peak Plasma (Cmax) | 45–60 minutes | 20–30 minutes | SubQ better matches endogenous melatonin secretion timing for circadian protocols |
| Duration Above Therapeutic Threshold | 4–6 hours | 90–120 minutes | SubQ provides sustained overnight coverage; IM fades before sleep cycle completes |
| Bioavailability (% absorbed) | 85–92% | 88–95% | Equivalent total absorption, but kinetic profile differs significantly |
| Injection Site Pain (VAS score) | 1.2–2.0 / 10 | 3.5–5.0 / 10 | IM causes 2.5–3× higher reported pain and tissue trauma |
| Tissue Trauma & Inflammation Risk | Low (hypodermis recovers quickly) | Moderate to High (fibrosis, hematoma, IL-6 elevation) | Repeated IM injections degrade muscle tissue and counteract anti-inflammatory effects |
| Suitability for Chronic Dosing | High (large rotation area, minimal scarring) | Low (limited muscle sites, cumulative trauma) | SubQ allows months of daily dosing without depot exhaustion |
| Professional Assessment | Preferred for sleep, circadian, and chronic neuroprotection studies | Use only for acute oxidative stress models requiring rapid peak concentration |
Key Takeaways
- Subcutaneous melatonin injection produces peak plasma concentration in 45–60 minutes with sustained therapeutic levels for 4–6 hours, closely mimicking natural pineal secretion patterns.
- Intramuscular injection delivers faster onset (20–30 minutes to peak) but causes 2.5–3× higher injection site pain and clears below therapeutic threshold within 90–120 minutes.
- Repeated IM injections trigger localized IL-6 and TNF-alpha elevation. Inflammatory markers that directly oppose melatonin's intended anti-inflammatory and antioxidant effects.
- SubQ administration reduces MT1/MT2 receptor desensitization in chronic dosing protocols by avoiding the supraphysiological Cmax spikes that trigger tachyphylaxis.
- For sleep latency and circadian entrainment research, SubQ consistently outperforms IM in maintaining efficacy beyond 14 days of consecutive dosing.
- Oil-based melatonin formulations injected IM can cause granuloma formation and altered absorption kinetics. A documented risk that aqueous SubQ injections avoid entirely.
What If: Melatonin Injection Scenarios
What If I Need Rapid Melatonin Onset for an Acute Oxidative Stress Model?
Use IM injection at the vastus lateralis with a 23-gauge, 1-inch needle. In rodent ischemia-reperfusion injury models or acute traumatic brain injury studies, the 20–30 minute time to peak allows melatonin to reach neuroprotective concentrations before secondary oxidative cascade peaks at 45–60 minutes post-injury. Accept the higher tissue trauma as a trade-off for timing precision. Limit IM use to single-dose or short-duration protocols (≤7 days) to avoid cumulative muscle damage that confounds injury recovery metrics.
What If My Protocol Requires Daily Melatonin Injections for 8–12 Weeks?
Switch to SubQ administration immediately and establish a rotation schedule across at least 6 distinct injection sites (lower abdomen left/right, lateral thigh left/right, upper arm left/right). Inject at the same time each day relative to the dark phase of your light cycle. Not at the same clock time. Monitor injection sites weekly for lipodystrophy or localized inflammation; if depot sites show tissue changes, expand rotation to additional areas. IM injection over this duration will cause fibrosis, altered pharmacokinetics, and elevated baseline cortisol from repeated pain exposure.
What If I'm Comparing Melatonin Routes in a Sleep Latency Study and Results Are Inconsistent?
Verify that IM and SubQ groups are being dosed at equivalent times relative to lights-out. A common error: injecting both groups 30 minutes before lights-out, which gives IM subjects peak melatonin at lights-out (ideal) but SubQ subjects peak melatonin 15–30 minutes after lights-out (suboptimal). Adjust SubQ injection timing to 60 minutes before lights-out to synchronize Cmax with sleep onset window. Inconsistent results between routes almost always trace back to this timing mismatch, not the route itself.
The Unvarnished Truth About Melatonin Injection Routes
Here's the honest answer: most researchers pick IM injection because it's what they learned in training. Not because the science supports it for melatonin specifically. The kinetic profile of IM melatonin is wrong for 80% of the research applications it's used in. If you're studying sleep, circadian entrainment, chronic neuroprotection, or metabolic signaling, IM gives you a sharp pharmacokinetic spike that looks impressive on a graph but doesn't match the biological process you're trying to influence. SubQ produces a boring, gradual curve. And that's exactly why it works better. The human circadian system evolved to respond to slow melatonin ramps, not bolus injections.
The tissue trauma argument alone should end the debate for chronic protocols. You can't run a 12-week study claiming melatonin reduces inflammation while simultaneously injecting it in a way that elevates IL-6 and causes muscle fibrosis. That's not rigorous. It's self-defeating experimental design. SubQ melatonin works. IM melatonin works faster but stops working sooner and damages tissue in the process. The choice should be obvious.
Technical Considerations for SubQ Melatonin Preparation
Melatonin for injection is typically prepared as a suspension in sterile saline, propylene glycol, or oil-based vehicles depending on solubility requirements and desired absorption kinetics. Aqueous melatonin solutions must be used immediately after preparation due to rapid oxidation and photodegradation. Store reconstituted melatonin in amber glass vials, refrigerate at 2–8°C, and use within 24–48 hours. Oil-based suspensions (common in veterinary research) extend stability to 7–14 days but require warming to body temperature before injection to reduce viscosity and injection site pain.
For SubQ administration, use a 25–27 gauge needle, 0.5–0.625 inches in length. Pinch the skin to create a subcutaneous tent, insert the needle at a 45-degree angle, and inject slowly over 3–5 seconds. Rapid injection increases local tissue pressure and pain scores. Rotate injection sites in a documented pattern to prevent lipodystrophy. Insulin protocols offer the most validated rotation schemes and translate directly to peptide administration.
Dose calculation must account for species-specific melatonin clearance rates. Rodents metabolize melatonin 5–8× faster than humans due to higher hepatic CYP1A2 expression, so doses in the 5–10 mg/kg range are common in mouse studies, while human equivalent doses rarely exceed 0.3 mg/kg. Always reference allometric scaling formulas when translating doses across species. Direct mg/kg conversion without body surface area adjustment is one of the most common errors in peptide research.
Our experience working with researchers at institutions running melatonin trials is that preparation errors cause more protocol failures than injection technique. If reconstituted melatonin sits on a benchtop under fluorescent light for 20 minutes, you've degraded 30–50% of the active compound before it ever reaches the subject. Light exposure, oxygen exposure, and pH drift all destroy melatonin rapidly. Treat it like a photosensitive compound. Because it is.
For teams looking to expand their peptide research toolkit beyond melatonin, understanding injection route pharmacokinetics is foundational. You can explore how similar principles apply to other research compounds like P21 and see how our commitment to quality extends across our full peptide collection.
The single biggest mistake researchers make with melatonin isn't the injection route. It's assuming all peptides behave the same way regardless of how they're delivered. They don't. Lipid solubility, molecular weight, receptor distribution, and clearance kinetics all determine whether SubQ or IM is the right choice. Melatonin happens to be a peptide where SubQ wins on nearly every metric that matters for the majority of research applications. That won't be true for every compound, but it's unambiguously true for this one.
Frequently Asked Questions
What is the main pharmacokinetic difference between SubQ and IM melatonin injection?
▼
Subcutaneous melatonin reaches peak plasma concentration in 45–60 minutes and maintains therapeutic levels for 4–6 hours due to slower absorption from adipose tissue. Intramuscular injection peaks faster (20–30 minutes) but clears below therapeutic threshold within 90–120 minutes because muscle tissue has higher capillary density and faster blood flow. The total bioavailability is similar (85–95% for both routes), but the shape of the concentration-time curve differs fundamentally — SubQ produces a sustained plateau, IM produces a sharp spike and rapid decline.
Why does IM injection cause more pain than SubQ for melatonin administration?
▼
Intramuscular injection requires a longer needle (1–1.5 inches) to penetrate through subcutaneous tissue and reach muscle, increasing the probability of hitting small blood vessels and nerve endings. Muscle tissue also contains more nociceptors (pain receptors) than the hypodermis. Clinical pain scores measured on the visual analog scale show IM injection produces 2.5–3× higher reported pain (3.5–5.0 / 10) compared to SubQ (1.2–2.0 / 10) across human, primate, and rodent studies.
Can I use IM injection for chronic melatonin protocols lasting several months?
▼
Not recommended — repeated IM injections into the same muscle group cause fibrosis, reduced capillary perfusion, and eventually altered absorption kinetics as scar tissue forms. Studies in laboratory animals show that daily IM injections beyond 3–4 weeks lead to measurable tissue damage and elevated baseline inflammatory markers (IL-6, TNF-alpha) that counteract melatonin’s anti-inflammatory effects. SubQ injection allows rotation across a much larger surface area with minimal depot site exhaustion, making it far superior for chronic dosing protocols.
Does the injection route affect melatonin’s effectiveness for sleep research?
▼
Yes — substantially. SubQ melatonin maintains plasma levels above the MT1 receptor activation threshold for 4–6 hours, matching the duration of the human sleep cycle and mimicking natural pineal secretion patterns. IM injection clears too quickly, often dropping below therapeutic levels within 90–120 minutes, which disrupts sustained circadian signaling. Sleep latency studies consistently show SubQ administration maintains efficacy longer in chronic dosing protocols (beyond 14 days) because it avoids the supraphysiological Cmax spikes that trigger MT1 receptor desensitization.
What needle size should I use for SubQ melatonin injection?
▼
Use a 25–27 gauge needle, 0.5–0.625 inches in length for subcutaneous administration. Insert at a 45-degree angle into pinched skin to create a subcutaneous tent, and inject slowly over 3–5 seconds to minimize tissue pressure and pain. For IM injection (if required for acute protocols), use a 23-gauge, 1-inch needle at the vastus lateralis or deltoid, inserted at a 90-degree angle to ensure penetration into muscle tissue.
How does injection route impact melatonin’s antioxidant effects?
▼
Supraphysiological melatonin concentrations produced by IM bolus injection can paradoxically increase oxidative stress in certain tissue contexts by overwhelming mitochondrial transport mechanisms and triggering excessive autophagy. SubQ administration keeps plasma melatonin within a narrower therapeutic window (typically 100–300 pg/mL sustained over hours), reducing the risk of this non-monotonic dose-response curve. For antioxidant research models, SubQ better replicates physiological melatonin levels and avoids the confounding effects of transient concentration spikes above 1,000 pg/mL.
What happens if I inject oil-based melatonin formulations intramuscularly?
▼
Oil-based melatonin injected IM can cause localized inflammation, granuloma formation, and prolonged absorption that extends half-life unpredictably — a documented adverse event in veterinary peptide studies. The viscosity of oil vehicles combined with the higher capillary density in muscle creates depots that don’t clear uniformly, leading to variable pharmacokinetics and potential chronic inflammatory nodules at injection sites. Oil-based formulations should be restricted to SubQ administration where slower lymphatic drainage is advantageous rather than problematic.
Why do some researchers still prefer IM injection despite the evidence favoring SubQ?
▼
Training inertia and visible pharmacokinetic curves. IM injection produces a sharp Cmax peak that looks dramatic on plasma concentration graphs, giving the subjective impression of ‘stronger’ efficacy even when total AUC (area under the curve) is equivalent to SubQ. Many researchers learned IM technique first in training and default to it without reviewing current pharmacokinetic literature specific to melatonin. The shift toward SubQ is driven by recognition that sustained receptor activation matters more than peak concentration for circadian and neuroprotective research.
Should I adjust melatonin injection timing differently for SubQ vs IM routes?
▼
Yes — critical for sleep studies. IM melatonin should be injected 20–30 minutes before the target onset time (e.g., lights-out in circadian protocols) to align Cmax with the desired physiological window. SubQ melatonin should be injected 60 minutes before the target onset to account for slower absorption kinetics. A common study design error is injecting both groups at the same pre-onset interval, which misaligns SubQ peak concentration with the biological process being measured and produces inconsistent results.
What is the maximum safe duration for daily melatonin injections using the SubQ route?
▼
SubQ melatonin can be administered daily for 12+ weeks in research protocols without depot site exhaustion if proper rotation schedules are followed (minimum 6 distinct injection sites rotated systematically). Clinical studies in humans and chronic dosing studies in rodents have documented continuous SubQ peptide administration for 6+ months without loss of absorption efficiency or significant tissue changes. The limiting factor is typically not the injection route but melatonin receptor downregulation in chronic high-dose protocols, which occurs regardless of administration method.