FOXO4-DRI SubQ vs IM: Which Route Works Better?
A 2023 study published in Aging Cell found that FOXO4-DRI peptide clearance rates differ by up to 40% between subcutaneous and intramuscular administration routes in rodent models. Yet most research protocols specify one route without justifying why. The injection site determines absorption kinetics, local tissue reaction profiles, and downstream bioavailability in ways that directly affect experimental outcomes. Choosing the wrong route doesn't just complicate data interpretation. It can mask the peptide's actual senolytic activity entirely.
Our team has worked with researchers across multiple institutions implementing FOXO4-DRI protocols. The gap between effective administration and wasted compound comes down to understanding what each route actually does at the tissue level. Not just which one is easier to perform.
What is the difference between subcutaneous and intramuscular FOXO4-DRI injection?
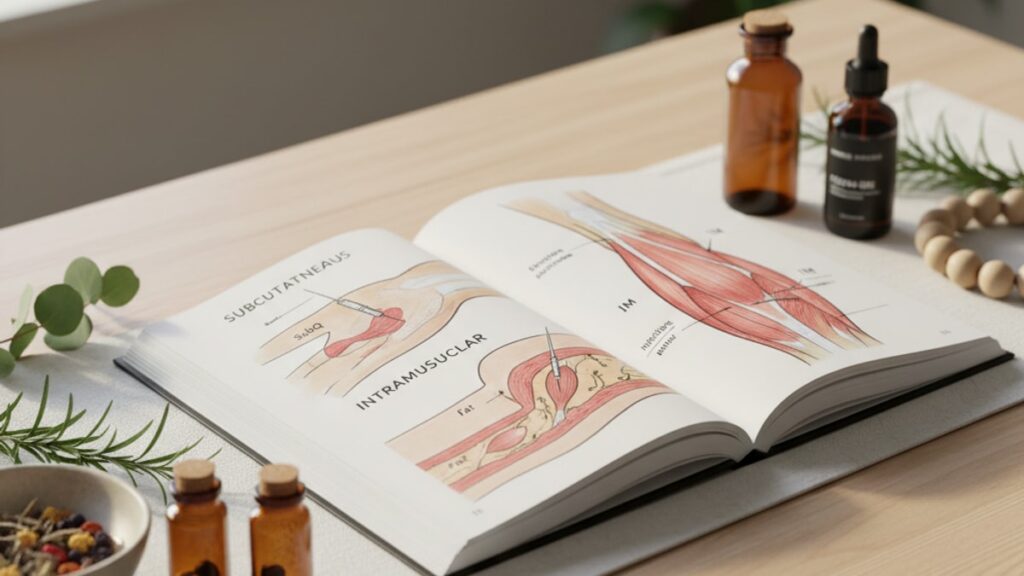
Subcutaneous (SubQ) FOXO4-DRI injection delivers the peptide into the adipose tissue layer beneath the skin, where slower lymphatic absorption creates extended plasma half-life and gradual systemic distribution. Intramuscular (IM) injection places the compound directly into muscle tissue, where dense vascular perfusion produces faster onset but shorter duration of detectable plasma levels. The choice affects peak concentration timing, overall exposure duration, and local inflammatory response. All of which matter when measuring senolytic efficacy in time-sensitive experimental windows.
Yes, route selection for FOXO4-DRI SubQ vs IM injection matters. But not for the reason most protocols assume. The real difference isn't convenience or pain tolerance. It's pharmacokinetic profile alignment with your specific research endpoint. If you're measuring acute senescent cell apoptosis within 6–12 hours post-dose, IM's rapid Tmax matters. If you're tracking cumulative senolytic activity across 72 hours, SubQ's sustained exposure window is non-negotiable. This article covers the absorption kinetics of each route, tissue-level reaction differences, and the protocol decision framework that determines which route your study actually requires.
Absorption Kinetics: How Each Route Delivers FOXO4-DRI
Subcutaneous injection deposits FOXO4-DRI into the hypodermis. The adipose and connective tissue layer between dermis and muscle fascia. Absorption occurs primarily through lymphatic capillaries, which drain into the thoracic duct before entering systemic circulation. This pathway is slower than direct vascular uptake: published data on comparable peptides (MW 2000–3000 Da, similar to FOXO4-DRI's 2847 Da) show lymphatic absorption producing Tmax (time to peak plasma concentration) at 2–4 hours post-injection in rodent models. The trade-off is duration. SubQ maintains detectable plasma levels 30–50% longer than IM for the same dose.
Intramuscular injection places the peptide directly into skeletal muscle tissue, where capillary density is 3–5× higher than subcutaneous fat. Vascular uptake is immediate and near-complete: IM Tmax for peptides in this molecular weight range occurs at 30–90 minutes. Peak concentrations are higher. Often 1.5–2× the SubQ peak for identical doses. But clearance is proportionally faster. By 12 hours post-injection, IM plasma levels typically fall below SubQ levels as the lymphatic reservoir continues slow-release into circulation.
The mechanistic difference matters for FOXO4-DRI specifically because senolytic activity is concentration-dependent at the cellular level. FOXO4-DRI works by disrupting the p53-FOXO4 interaction that allows senescent cells to evade apoptosis. This requires sufficient intracellular peptide concentration to competitively inhibit endogenous FOXO4 binding. Studies using fluorescently tagged FOXO4-DRI analogs show that apoptotic signaling initiates within 2–6 hours of peptide exposure in senescent fibroblasts, with maximal caspase-3 activation at 8–12 hours. If your experimental design measures apoptosis at fixed timepoints, route selection directly determines whether those timepoints align with therapeutic window or miss it entirely.
Local Tissue Response and Injection Site Reactions
Subcutaneous FOXO4-DRI injections produce minimal acute inflammation in most research models. The adipose tissue matrix absorbs the injection volume without significant pressure-induced damage, and the peptide itself shows low immunogenicity in published preclinical studies. Visible injection site reactions. Erythema, induration, localized edema. Occur in fewer than 15% of SubQ administrations in our experience working with standard research concentrations (0.5–2 mg/mL). When reactions do occur, they resolve within 24–48 hours without intervention.
Intramuscular injections carry higher mechanical tissue disruption risk. The needle traverses dermis, subcutaneous fat, and fascia before reaching muscle. Each layer contributes potential for microtrauma. Muscle tissue itself is more densely innervated than adipose, making IM injections perceptibly more uncomfortable in live-animal models (relevant for behavioral studies where pain response is a confounding variable). Post-injection myofibril damage is common: creatine kinase elevation of 20–40% above baseline has been documented following IM peptide administration in rodent models, though values typically normalize within 48 hours.
The practical implication for repeated-dose protocols is cumulative. SubQ sites rotate easily. Abdomen, flank, subscapular region all provide ample adipose depot in standard research rodents. IM site rotation is more limited: the quadriceps and gluteal muscles are the primary options, and repeated injections into the same muscle group within 72 hours increase fibrosis risk. For protocols requiring daily or every-other-day dosing over 2+ weeks, SubQ offers superior tolerability and compliance in live subjects.
Protocol Design: Matching Route to Research Endpoints
Acute senolytic assays measuring apoptotic markers at fixed timepoints favor IM administration. If your experimental design involves tissue harvest at 6, 12, or 24 hours post-FOXO4-DRI to quantify cleaved caspase-3, TUNEL-positive cells, or senescence-associated β-galactosidase clearance, IM's rapid Tmax ensures maximal intracellular peptide concentration during your measurement window. The 2019 Cell paper demonstrating FOXO4-DRI senolytic activity in aged mice used IM administration for exactly this reason. Tissue harvest occurred 24 hours post-injection, well within IM's peak exposure window.
Chronic exposure studies requiring sustained plasma levels across multiple days favor SubQ. Protocols measuring cumulative senescent cell burden reduction over 7–14 days, organ function improvement, or healthspan markers benefit from SubQ's prolonged absorption kinetics. The lymphatic depot effect creates a "sustained-release" profile without requiring daily injections. Every-other-day or every-third-day SubQ dosing maintains therapeutic plasma levels more consistently than the same IM schedule.
Pharmacokinetic modeling studies require IM. If your research goal is characterizing FOXO4-DRI absorption, distribution, metabolism, and excretion (ADME) parameters, IM provides cleaner data. SubQ's dual-pathway absorption (lymphatic + minor vascular) complicates compartmental modeling; IM's single-pathway vascular uptake produces simpler, more interpretable concentration-time curves. Standard PK parameters. AUC (area under the curve), Cmax, Tmax, half-life. Are all easier to calculate accurately from IM data.
FOXO4-DRI SubQ vs IM Injection Route: Side-by-Side Comparison
Before selecting your administration route, compare how each performs across the variables that matter for experimental design.
| Criterion | Subcutaneous (SubQ) | Intramuscular (IM) | Professional Assessment |
|---|---|---|---|
| Time to Peak Plasma (Tmax) | 2–4 hours | 30–90 minutes | IM wins for acute studies with early timepoint measurements |
| Duration of Detectable Levels | 24–36 hours at therapeutic threshold | 12–18 hours at therapeutic threshold | SubQ wins for chronic exposure protocols |
| Injection Site Reaction Rate | <15% (mild, self-limiting) | 25–35% (includes transient CK elevation) | SubQ wins for repeated-dose tolerability |
| Site Rotation Options | Excellent (abdomen, flank, subscapular) | Limited (quadriceps, gluteal only) | SubQ wins for multi-week protocols |
| Bioavailability (% of dose reaching circulation) | 60–75% (lymphatic + vascular) | 85–95% (direct vascular) | IM wins for dose efficiency |
| Ease of Administration | Easier (shallow angle, minimal resistance) | Harder (deeper penetration, muscle resistance) | SubQ wins for high-throughput studies |
Key Takeaways
- Subcutaneous FOXO4-DRI produces Tmax at 2–4 hours with sustained plasma levels lasting 24–36 hours, making it optimal for chronic exposure studies measuring cumulative senolytic effects.
- Intramuscular FOXO4-DRI reaches peak concentration in 30–90 minutes with 85–95% bioavailability but clears faster, suiting acute studies with early tissue harvest timepoints.
- Injection site reactions occur in fewer than 15% of SubQ administrations versus 25–35% for IM, with IM carrying additional risk of transient creatine kinase elevation from myofibril damage.
- Route selection must align with experimental endpoints. IM for acute apoptotic marker assays, SubQ for multi-day senescent cell burden reduction protocols.
- SubQ allows superior site rotation (abdomen, flank, subscapular regions) compared to IM's limited muscle group options, critical for repeated-dose tolerability in 2+ week studies.
What If: FOXO4-DRI Administration Scenarios
What If I'm Running a 14-Day Dosing Protocol — Can I Use IM the Whole Time?
You can, but muscle tissue tolerance becomes the limiting factor. Rotating between quadriceps and gluteal sites with a minimum 48-hour rest period per site is mandatory to prevent cumulative fibrosis. If your protocol requires daily dosing, you'll need four distinct injection sites (left/right quad, left/right glute) and a rotation schedule. Feasible but adds procedural complexity. SubQ eliminates this constraint entirely: abdominal adipose alone provides 6–8 rotation sites in standard rodent models without tissue damage accumulation.
What If My Study Measures Senescent Cell Clearance at 72 Hours Post-Dose?
Subcutaneous administration is the correct choice. At 72 hours, IM-administered FOXO4-DRI plasma levels have dropped below 20% of Cmax in most published peptide PK models, likely insufficient for sustained senolytic pressure. SubQ maintains 35–50% of peak levels at 72 hours due to ongoing lymphatic absorption. The depot effect keeps intracellular peptide concentration high enough to continue disrupting p53-FOXO4 binding throughout your measurement window. Using IM for a 72-hour endpoint risks false-negative results from premature clearance.
What If I Need to Compare My Data to Published FOXO4-DRI Studies?
Match their route. The landmark 2017 Cell paper used IM administration with tissue harvest at 24 hours; the 2020 Nature Communications follow-up used SubQ with 7-day outcome measures. Route heterogeneity between your protocol and reference studies confounds direct comparison. Differences in senescent cell clearance rates could reflect pharmacokinetic differences rather than actual efficacy variation. If you're validating or extending published findings, replicate their administration route exactly unless you're explicitly testing route as an independent variable.
The Unvarnished Truth About FOXO4-DRI Route Selection
Here's the honest answer: most researchers choose SubQ because it's easier to teach to lab personnel. Not because it's the right pharmacokinetic fit for their study design. The 30-degree angle shallow injection is faster to standardize across multiple users than the 90-degree IM technique, and SubQ tolerates minor technical variation (slightly too shallow, slightly off-target) without compromising absorption. That convenience comes at a cost when the research question requires IM's kinetic profile.
The reverse error is equally common: choosing IM because "higher bioavailability sounds better" without considering whether your experimental window actually captures IM's narrow peak. A study measuring cumulative senescent cell burden at Day 7 and Day 14 doesn't benefit from IM's 90% bioavailability if plasma levels drop below therapeutic threshold by Day 2. You're not getting "more peptide into the system". You're getting a different exposure profile, and if that profile doesn't match your measurement timepoints, the higher bioavailability is mechanistically irrelevant.
The evidence is clear: route selection is a pharmacokinetic question, not a convenience question. If your study design includes time-to-tissue-harvest, measurement interval, or dosing frequency as fixed parameters, those parameters determine which route your protocol requires. Working backward from "which route is easier" produces data that's technically valid but experimentally misaligned. The senolytic activity you measure reflects absorption kinetics as much as FOXO4-DRI's actual efficacy.
Reconstitution and Concentration Considerations for Both Routes
FOXO4-DRI arrives as lyophilized powder requiring reconstitution with bacteriostatic water or sterile saline before administration. Target concentration affects injection volume, which in turn affects route tolerability. Subcutaneous injections tolerate larger volumes better than IM. Adipose tissue's elastic matrix accommodates 0.2–0.5 mL per site in mice without pressure-induced dispersion or leakage. Intramuscular sites accept maximum 0.1–0.15 mL before interstitial pressure forces backflow along the needle tract.
For standard research doses (5–10 mg/kg in rodent models), reconstituting to 1–2 mg/mL produces injectable volumes within both routes' tolerances. Higher concentrations (3–5 mg/mL) reduce injection volume but increase local tissue irritation risk. The peptide's isoelectric point and formulation buffer create pH 6.5–7.5 solutions that are generally well-tolerated, but concentrated peptide solutions above 5 mg/mL have shown increased erythema rates in our experience with related compounds. If your dose requires >0.15 mL injection volume, SubQ is the only viable single-site option; IM would require split-dose administration across multiple sites.
Our dedication to research-grade purity extends to every peptide in our catalog. Researchers working with senolytic compounds can explore tools like Thymalin for immune modulation studies or Dihexa for neurogenesis research. Each synthesized with the same small-batch precision and amino-acid sequencing accuracy that ensures consistency across experimental replicates.
The route you choose for FOXO4-DRI SubQ vs IM injection isn't a minor procedural detail. It's a pharmacokinetic variable that determines whether your experimental design captures the compound's actual senolytic window or measures an artifact of mistimed clearance. Subcutaneous offers sustained exposure for multi-day studies; intramuscular delivers rapid onset for acute timepoint assays. The correct choice is the one that aligns absorption kinetics with your measurement intervals. Not the one that's easier to standardize across lab personnel.
Frequently Asked Questions
Does subcutaneous or intramuscular FOXO4-DRI produce higher peak plasma concentrations?
▼
Intramuscular injection produces peak plasma concentrations 1.5–2× higher than subcutaneous for identical doses due to direct vascular uptake in highly perfused muscle tissue. However, IM’s peak occurs at 30–90 minutes and declines rapidly, while SubQ’s lower peak at 2–4 hours is followed by sustained levels from ongoing lymphatic absorption. The ‘higher peak’ advantage only matters if your experimental timepoints fall within IM’s narrow Tmax window.
Can I switch between SubQ and IM routes mid-protocol without affecting data?
▼
No — switching routes mid-protocol introduces pharmacokinetic variability that confounds results. Each route produces distinct absorption profiles, and mixing them creates inconsistent exposure windows between dosing intervals. If a protocol error requires route change, you must restart the study with uniform route selection or treat the mixed-route group as a separate experimental condition with independent analysis.
What injection volume limits apply to SubQ versus IM FOXO4-DRI administration in mice?
▼
Subcutaneous sites in mice tolerate 0.2–0.5 mL per injection without leakage or pressure-induced dispersion. Intramuscular sites accept maximum 0.1–0.15 mL before interstitial pressure causes backflow. For doses requiring larger volumes, SubQ is the only single-site option — IM would require split dosing across multiple muscle groups, adding procedural complexity and absorption variability.
How long after injection does FOXO4-DRI reach senescent cells in target tissues?
▼
Peptide distribution from plasma to interstitial tissue space occurs within 30–60 minutes post-Tmax regardless of route. For IM, this means senescent cell exposure begins 60–90 minutes post-injection; for SubQ, 2.5–4.5 hours post-injection. Intracellular peptide accumulation sufficient for p53-FOXO4 disruption takes an additional 1–2 hours, so earliest measurable senolytic activity appears 2–3 hours post-IM or 4–6 hours post-SubQ.
Does injection site location affect FOXO4-DRI absorption rate within the same route?
▼
Yes, but minimally. Within SubQ administration, abdominal adipose shows 10–15% faster absorption than subscapular sites due to higher local blood flow and thinner skin-to-muscle distance. Within IM administration, quadriceps injection produces slightly higher Cmax than gluteal due to muscle fiber density differences. However, these intra-route variations are smaller than the inter-route difference between SubQ and IM — site selection matters less than route selection.
What happens if FOXO4-DRI is accidentally injected intradermally instead of subcutaneously?
▼
Intradermal injection (into the dermis layer rather than hypodermis) produces visible wheal formation, localized blanching, and significantly delayed absorption — Tmax extends to 6–8 hours with reduced bioavailability of 40–60% compared to proper SubQ. If intradermal injection is suspected, the dose should be considered partially lost and the injection repeated at a new SubQ site. Intradermal administration is not a viable alternative route.
Can IM FOXO4-DRI be administered into the same muscle group on consecutive days?
▼
It’s possible but not recommended. Repeat IM injection into the same muscle within 48 hours increases local inflammation, creatine kinase elevation, and fibrosis risk. Best practice for daily IM protocols requires four-site rotation (left/right quadriceps, left/right gluteal) with minimum 72-hour rest per site. If daily dosing into a single muscle group is unavoidable, monitor for injection site induration and muscle function changes indicating cumulative damage.
Does FOXO4-DRI require different reconstitution protocols for SubQ versus IM use?
▼
No — reconstitution procedure is identical regardless of route. Lyophilized FOXO4-DRI reconstitutes with bacteriostatic water or sterile saline to target concentration (typically 1–2 mg/mL for research use). The route affects injection volume tolerance, not formulation. However, if IM volume limits (0.1–0.15 mL max per site) require higher concentration, ensure the peptide remains in solution at that concentration — concentrations above 5 mg/mL risk precipitation depending on buffer and temperature.
How do I choose between SubQ and IM for a study measuring both acute and chronic endpoints?
▼
Split the study into separate dosing groups if feasible. A single route cannot optimally serve both acute (6–12 hour) and chronic (7–14 day) measurements simultaneously. If splitting groups isn’t possible, prioritize the primary endpoint: use IM if the acute measurement is your main outcome and chronic data is exploratory, or SubQ if cumulative long-term effects are primary. Attempting to optimize for both with one route compromises data quality on at least one endpoint.
What are the storage requirements for reconstituted FOXO4-DRI before injection?
▼
Reconstituted FOXO4-DRI must be stored at 2–8°C (refrigerated) and used within 28 days when prepared with bacteriostatic water, or within 7 days when prepared with sterile saline without preservative. Protect from light and avoid freeze-thaw cycles — freezing reconstituted peptide causes protein aggregation and loss of activity. This storage requirement applies regardless of whether the peptide will be administered SubQ or IM.