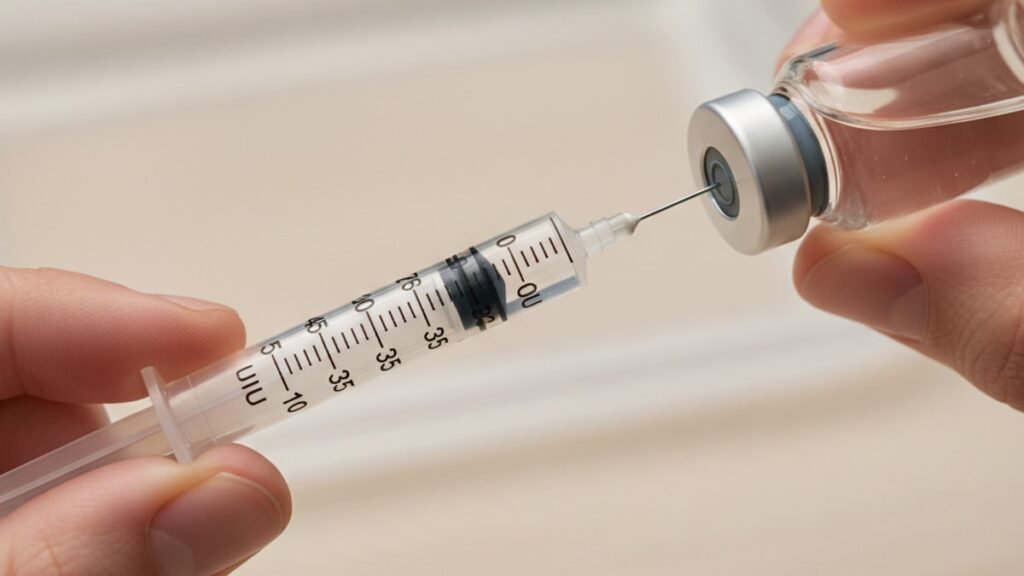
Insulin Syringes for Peptides: Unit Conversion Guide
Most reconstituted peptide dosing errors don't happen during mixing. They happen during injection because insulin syringes use unit markings, not milliliters, and one misread line can deliver ten times the intended dose. A 0.5mL insulin syringe marked to 50 units is calibrated for U-100 insulin (100 units per milliliter). Not for arbitrary peptide concentrations. Drawing to the "25 unit" line delivers 0.25mL of solution, regardless of what peptide is dissolved in that volume.
We've walked hundreds of researchers through this exact conversion process at Real Peptides. The mistakes are always the same: assuming the syringe scale matches the peptide concentration, miscounting tick marks under poor lighting, or forgetting that "units" on the barrel refer to insulin units. Not micrograms of peptide. This guide covers the exact math, the syringe types that prevent errors, and the reconstitution mistakes that make accurate dosing impossible.
How do you convert peptide doses to insulin syringe units?
Insulin syringe units measure volume, not peptide mass. If you reconstitute 5mg of peptide in 2mL of bacteriostatic water, your concentration is 2.5mg/mL. Drawing to the 20-unit mark on a U-100 syringe delivers 0.2mL of solution, which contains 0.5mg of peptide. The conversion formula is: (desired dose in mg ÷ concentration in mg/mL) × 100 = units to draw. Errors occur when researchers treat syringe units as direct peptide measurements rather than volume markers.
Why Standard Insulin Syringes Fail for Peptide Research
Insulin syringes are engineered for one specific use case: delivering pharmaceutical insulin at a standardized concentration of 100 units per milliliter. Every marking on the barrel corresponds to that fixed ratio. 10 units equals 0.1mL, 50 units equals 0.5mL, 100 units equals 1.0mL. This works flawlessly for insulin because the concentration never changes. Peptide research operates under completely different parameters: lyophilized peptides arrive as dry powder at arbitrary masses (2mg, 5mg, 10mg, 20mg) and must be reconstituted with bacteriostatic water at researcher-determined volumes.
The problem emerges immediately after reconstitution. A 5mg vial of CJC1295 Ipamorelin reconstituted with 2mL of bacteriostatic water produces a concentration of 2.5mg/mL. Nowhere near the 100 units/mL insulin standard. Drawing to the "20 unit" line on your syringe delivers 0.2mL of solution, which at 2.5mg/mL concentration contains 0.5mg of peptide. The syringe marking says "20 units". But you're not dosing 20 units of anything. You're dosing 0.2 milliliters of a custom-concentration solution.
Research facilities that process multiple peptides daily see this error pattern repeatedly: technicians who reconstitute correctly but dose incorrectly because they're reading unit markings as peptide quantities. A Tesofensine protocol calling for 250mcg per administration becomes 2.5mg when the technician draws to "25 units" without converting volume to concentration first. The ten-fold overdose isn't a calculation error. It's a misunderstanding of what the syringe measures.
The Three-Step Conversion Formula That Prevents Dosing Errors
Every peptide dose requires three known values before you can determine where to draw on an insulin syringe: peptide mass in the vial, reconstitution volume, and target dose. Missing any one value makes accurate dosing impossible. The calculation sequence runs in this exact order.
Step 1: Calculate concentration. Divide total peptide mass by reconstitution volume. A 10mg vial of Hexarelin reconstituted with 2mL of bacteriostatic water produces 5mg/mL concentration. This number becomes the foundation for every subsequent dose.
Step 2: Convert target dose to volume. Divide your desired dose in milligrams by the concentration in mg/mL. If your protocol calls for 200mcg (0.2mg) of Hexarelin and your concentration is 5mg/mL, the required volume is 0.2mg ÷ 5mg/mL = 0.04mL. This is the physical volume of liquid you need to draw. Not a syringe unit reading.
Step 3: Convert milliliters to syringe units. Multiply the required volume in milliliters by 100. Since insulin syringes are calibrated to U-100 standard (100 units = 1mL), 0.04mL × 100 = 4 units on the syringe barrel. Draw to the 4-unit line and you've delivered exactly 0.2mg of peptide.
The formula condenses to: (target dose in mg ÷ concentration in mg/mL) × 100 = units to draw. For researchers working with consistent protocols, pre-calculating a conversion table eliminates real-time math errors. If you're administering MK 677 at 10mg/dose from a 50mg vial reconstituted with 5mL bacteriostatic water, your concentration is 10mg/mL. Every 10-unit draw delivers exactly 1mg, so your 10mg dose requires drawing to the 100-unit line (1.0mL). Document this once and reference it for every subsequent administration.
How Reconstitution Volume Determines Syringe Precision Limits
Insulin syringes come in three standard volumes: 0.3mL (30 units), 0.5mL (50 units), and 1.0mL (100 units). Each size has different tick-mark graduations that dictate the smallest measurable dose. A 1mL syringe typically has 1-unit graduations. You can measure 0.01mL increments with reasonable accuracy. A 0.3mL syringe often has 0.5-unit or 1-unit graduations depending on manufacturer, limiting your precision to 0.005mL or 0.01mL respectively.
This creates a binding constraint: your reconstitution volume must produce a concentration that allows your target dose to fall on a readable graduation line. Researchers dosing Dihexa at 5mg per administration from a 50mg vial face a decision point. Reconstituting with 10mL of bacteriostatic water produces 5mg/mL concentration. A 5mg dose requires 1.0mL (100 units on the syringe), which is readable but uses the entire barrel of a 1mL syringe with no margin for air bubbles or measurement error.
Reconstituting the same vial with 5mL instead produces 10mg/mL concentration. Now a 5mg dose requires only 0.5mL (50 units), leaving half the barrel empty and providing visual confirmation that you've drawn correctly. The tradeoff: higher concentration means smaller volumes for lower doses, which can fall below the precision threshold of your syringe. A 500mcg dose at 10mg/mL concentration requires 0.05mL (5 units). Perfectly measurable on a 1mL syringe with 1-unit graduations, but difficult to read accurately on a 0.5mL syringe with 2-unit graduations.
Our experience managing peptide protocols at Real Peptides shows that most researchers optimize for mid-range syringe readings. Targeting doses that fall between 10 and 50 units on the barrel, where parallax error and meniscus misreading have minimal impact. A Cartalax protocol calling for 2mg daily from a 20mg vial works cleanly with 2mL reconstitution volume: concentration becomes 10mg/mL, and the 2mg dose requires 0.2mL (20 units). A mid-barrel draw with clear visual confirmation.
Comparison Table: Insulin Syringe Types for Peptide Administration
| Syringe Size | Graduation | Best Use Case | Precision Limit | Typical Error Source | Professional Assessment |
|---|---|---|---|---|---|
| 0.3mL (30 units) | 1 unit (0.01mL) | Low-volume peptides requiring <0.3mL per dose; ideal for high-concentration solutions | ±0.01mL | Difficult to see graduations; needle too short for some injection sites | Best for researchers working with concentrated solutions and small doses. But only if your protocol never exceeds 0.3mL per administration |
| 0.5mL (50 units) | 1 unit (0.01mL) | Mid-range dosing for peptides requiring 0.1–0.4mL per administration | ±0.01mL | Parallax misreading at barrel extremes; air bubble displacement | The most versatile choice for peptide research. Handles most reconstitution volumes without hitting ceiling or floor limits |
| 1.0mL (100 units) | 1 unit (0.01mL) or 2 units (0.02mL) | High-volume peptides or low-concentration solutions requiring up to 1.0mL per dose | ±0.01–0.02mL depending on graduations | Meniscus reading errors in upper barrel; wasted solution if dose is consistently <0.5mL | Use when your protocol requires >0.5mL per dose. Otherwise you're using an oversized tool that introduces unnecessary measurement error |
Key Takeaways
- Insulin syringe "units" measure volume at U-100 insulin concentration (100 units = 1mL), not peptide mass. Drawing to the 50-unit line delivers 0.5mL of solution regardless of what's dissolved in it.
- The conversion formula is: (target dose in mg ÷ concentration in mg/mL) × 100 = syringe units to draw.
- Reconstitution volume determines both your concentration and your syringe precision limits. A 5mg peptide reconstituted with 1mL yields 5mg/mL, requiring 0.2mL (20 units) for a 1mg dose.
- Standard 1mL insulin syringes have 1-unit graduations (0.01mL precision), while 0.5mL syringes offer the same precision in a smaller barrel suited for doses under 0.5mL.
- Pre-calculating a dose-to-units conversion table for your specific peptide and reconstitution volume eliminates real-time math errors during administration.
- The most common error isn't miscalculation. It's treating syringe unit markings as direct peptide measurements rather than volume indicators.
What If: Insulin Syringe Peptide Dosing Scenarios
What If My Peptide Protocol Requires a Dose Smaller Than 0.01mL?
Increase your reconstitution volume to dilute the concentration. If you're dosing KPV at 200mcg per administration from a 5mg vial and reconstitute with 1mL of bacteriostatic water, your concentration is 5mg/mL. The required 0.2mg dose converts to 0.04mL (4 units), which sits at the edge of reliable measurement on a standard insulin syringe. Reconstituting with 2mL instead drops concentration to 2.5mg/mL, and your 200mcg dose now requires 0.08mL (8 units). Double the syringe reading, cutting measurement error in half.
What If I Accidentally Draw Air Into the Syringe While Measuring?
Expel it immediately before injecting. Air bubbles displace solution volume and cause underdosing. A 0.02mL air bubble in a 0.2mL dose (20-unit draw) represents 10% underdosing. Hold the syringe vertically with the needle pointing up, tap the barrel to collect bubbles at the top, then push the plunger slowly until liquid reaches the needle hub with no visible air remaining. Redraw to your target unit marking if solution was expelled during bubble removal.
What If My Syringe Has 2-Unit Graduations Instead of 1-Unit?
Round your calculated dose to the nearest even number on the barrel. If your protocol calls for 0.15mL (15 units) but your syringe only marks 14 and 16, draw to 16 units. The 0.01mL difference (1% variance on a typical 10mg peptide dose) falls within acceptable research margins. For protocols requiring tighter precision, switch to a 1mL syringe with 1-unit graduations or increase reconstitution volume to shift your target dose onto an even graduation line.
What If I Reconstituted at the Wrong Volume and Already Started Dosing?
Recalculate your concentration based on actual reconstitution volume and determine how much peptide you've already administered. If you intended to reconstitute a 10mg Thymalin vial with 2mL but used 1mL instead, your concentration is 10mg/mL rather than the intended 5mg/mL. Every dose you thought was 1mg actually delivered 2mg. Document the error, calculate remaining peptide in the vial, and either continue at the higher concentration with adjusted syringe readings or reconstitute a fresh vial at the correct volume.
The Unspoken Truth About Peptide Dosing Errors
Here's what most peptide suppliers won't tell you directly: the majority of "underdosed" or "bunk" peptide complaints trace back to user reconstitution and dosing errors, not product purity. When a researcher reports that a 5mg vial of Cerebrolysin "didn't work," the first question isn't about the peptide. It's about their reconstitution volume, their syringe type, and whether they understood that insulin syringe units aren't peptide units.
We've reviewed dosing protocols from hundreds of research teams. The pattern is consistent: researchers who document their concentration, verify their syringe graduations, and double-check their unit-to-volume conversion before every draw report consistent results. Researchers who eyeball the syringe reading or assume "25 units" on the barrel equals 25mg of peptide report erratic outcomes. Not because the peptide quality varies, but because their actual administered dose varies by 200–300% from one injection to the next. The difference between effective research and wasted product almost always comes down to whether the researcher treated dosing as a multi-step calculation requiring verification or as a visual approximation that "looks about right."
If you're transitioning from pre-filled pharmaceutical delivery systems to reconstituted research peptides, the learning curve isn't optional. Insulin syringes are precision instruments when used correctly and catastrophic when misunderstood. Every peptide protocol at Real Peptides includes reconstitution instructions, target concentrations, and dose-to-units conversion tables specifically because we've seen what happens when researchers skip this step. The $200 you spend on a botched vial isn't the peptide's fault. It's the cost of assuming measurement tools work the way you think they do rather than the way they're actually calibrated.
Misreading syringe units as peptide mass is the single most expensive mistake in research peptide administration. The fix is procedural: calculate concentration, convert dose to volume, multiply by 100, and draw to that unit marking. Do this every time. Document it every time. The consistency of your research outcomes depends on it.
Why Bacteriostatic Water Volume Matters More Than Peptide Mass
The mass of peptide in your vial is fixed. You can't change the 5mg, 10mg, or 20mg printed on the label. The variable you control entirely is reconstitution volume, and that single decision determines whether your dosing will be precise or guesswork. Researchers often default to "standard" reconstitution volumes without considering how that volume interacts with their target dose and their syringe's graduation limits.
A 10mg vial of P21 can be reconstituted with 1mL, 2mL, 5mL, or 10mL of bacteriostatic water. Each choice produces a different concentration and requires different syringe readings for the same dose. If your protocol calls for 500mcg (0.5mg) per administration, here's how volume choice affects measurement:
- 1mL reconstitution → 10mg/mL concentration → 0.5mg dose requires 0.05mL (5 units)
- 2mL reconstitution → 5mg/mL concentration → 0.5mg dose requires 0.1mL (10 units)
- 5mL reconstitution → 2mg/mL concentration → 0.5mg dose requires 0.25mL (25 units)
- 10mL reconstitution → 1mg/mL concentration → 0.5mg dose requires 0.5mL (50 units)
All four scenarios deliver the identical 500mcg dose. The difference is measurement precision and margin for error. Drawing to the 5-unit line on a 1mL syringe sits at the bottom of the barrel where meniscus reading is difficult and a single-unit mismeasurement represents 20% dose variance. Drawing to the 25-unit line puts you in the middle of the barrel with clear visual confirmation and a 1-unit error representing only 4% variance. The peptide quality is identical across all four. The reliability of your dose administration isn't.
Our team at Real Peptides recommends targeting syringe readings between 10 and 50 units whenever protocol flexibility allows. This range maximizes readability, minimizes parallax error, and keeps your dose in the "sweet spot" where graduation lines are easiest to distinguish. Calculate backward from your target dose to determine optimal reconstitution volume: if you need 1mg per administration from a 20mg vial, reconstituting with 2mL yields 10mg/mL concentration and requires 0.1mL (10 units) per dose. A clean mid-barrel reading. Reconstituting with 4mL drops concentration to 5mg/mL and requires 0.2mL (20 units). An even cleaner reading with more room for visual verification.
Bacteriostatic water is stable for 28 days after opening when refrigerated at 2–8°C. Your reconstitution volume choice should account for how many doses you'll administer within that window. A 50mg vial reconstituted with 5mL yields 10mg/mL. If you're dosing 5mg per day, that's 10 doses over 10 days, well within the stability window. The same vial reconstituted with 10mL yields 5mg/mL and requires 20 doses at the same daily intake. You'd need to discard remaining solution after 28 days regardless of whether peptide remains. Choose reconstitution volume based on your administration schedule, not arbitrary "standard" volumes that may not match your protocol.
The information in this article is for research and educational purposes. All peptide handling, reconstitution, and administration decisions should follow institutional protocols and be conducted by trained personnel in appropriate laboratory settings. For high-purity research-grade peptides formulated with exact amino-acid sequencing and supplied with detailed reconstitution guidance, explore our full peptide collection.
Frequently Asked Questions
How do you calculate how many units to draw on an insulin syringe for peptide dosing?
▼
Divide your target dose in milligrams by your concentration in mg/mL, then multiply by 100. If you reconstituted a 10mg peptide vial with 2mL of bacteriostatic water, your concentration is 5mg/mL — a 1mg dose requires (1mg ÷ 5mg/mL) × 100 = 20 units on the syringe. The syringe measures volume, not peptide mass, so you must convert your dose through concentration first.
Can you use a regular insulin syringe for reconstituted peptides?
▼
Yes, insulin syringes are the standard tool for subcutaneous peptide administration because they deliver precise volumes in the 0.01–1.0mL range and use fine-gauge needles (typically 29G–31G) that minimize tissue trauma. The critical requirement is understanding that syringe ‘units’ measure volume at U-100 insulin concentration (100 units = 1mL), not peptide quantity — you must calculate the volume of your custom-concentration peptide solution and draw to the corresponding unit marking.
What size insulin syringe is best for peptide injections?
▼
A 0.5mL (50-unit) insulin syringe with 1-unit graduations handles most peptide protocols efficiently — it provides 0.01mL precision across a range that accommodates doses from 0.05mL to 0.5mL without using extreme ends of the barrel where measurement error increases. Use a 1mL syringe only when your protocol consistently requires doses above 0.5mL; use a 0.3mL syringe when working with highly concentrated solutions where doses never exceed 0.3mL.
How much bacteriostatic water should I use to reconstitute peptides for insulin syringe dosing?
▼
Choose reconstitution volume so your target dose falls between 10 and 50 units on the syringe barrel — this range maximizes measurement precision and visual confirmation. For a 10mg peptide vial with a 500mcg daily dose, reconstituting with 2mL yields 5mg/mL concentration and requires 10 units per dose; reconstituting with 5mL yields 2mg/mL and requires 25 units per dose. Both deliver identical peptide amounts, but the 25-unit reading is easier to measure accurately.
What happens if you use the wrong reconstitution volume for peptide dosing?
▼
Using an unintended reconstitution volume changes your concentration, which changes the syringe units required for your target dose — if you’re following a protocol written for a different volume, you’ll overdose or underdose unless you recalculate. A 5mg vial reconstituted with 1mL instead of 2mL doubles your concentration from 2.5mg/mL to 5mg/mL — drawing to ’20 units’ delivers 1mg instead of the intended 0.5mg. Always calculate concentration from actual volume used, not protocol assumptions.
Are insulin syringe units the same as micrograms of peptide?
▼
No — insulin syringe units measure liquid volume, not peptide mass. One unit on a U-100 insulin syringe equals 0.01mL of solution, and the amount of peptide in that 0.01mL depends entirely on your reconstitution concentration. At 5mg/mL concentration, 10 units (0.1mL) contains 0.5mg of peptide; at 10mg/mL concentration, the same 10-unit volume contains 1mg. Never treat unit markings as direct peptide measurements.
How do you avoid air bubbles when drawing peptides into an insulin syringe?
▼
Hold the vial inverted with the needle submerged fully in solution, draw slowly to minimize turbulence, then hold the filled syringe vertically with the needle pointing up and tap the barrel to collect bubbles at the top. Push the plunger slowly until solution reaches the needle hub with no visible air — this expels the bubble without losing significant volume. Redraw to your target unit marking if you expelled enough liquid to drop below your intended dose.
Can you reuse insulin syringes for multiple peptide doses?
▼
No — insulin syringes are single-use sterile devices, and reusing them introduces contamination risk that can degrade your peptide solution or cause injection-site infection. The needle dulls after the first puncture (both through the vial stopper and tissue), increasing tissue trauma and reducing measurement accuracy. For research-grade peptide work, use a fresh sterile syringe for every administration.
What is the difference between U-100 and U-40 insulin syringes for peptide dosing?
▼
U-100 syringes are calibrated so 100 units equals 1.0mL; U-40 syringes are calibrated so 40 units equals 1.0mL — using the wrong type causes dose errors even if your volume calculation is correct. U-100 is the overwhelming standard in research settings and all conversion formulas assume U-100 calibration. If you accidentally use a U-40 syringe and draw to ’20 units,’ you’ve delivered 0.5mL instead of 0.2mL — a 2.5× overdose.
How long does reconstituted peptide remain stable in bacteriostatic water?
▼
Most lyophilized peptides remain stable for 28 days after reconstitution with bacteriostatic water when stored at 2–8°C in a refrigerator. Bacteriostatic water contains 0.9% benzyl alcohol as a preservative, which inhibits bacterial growth during that window. Beyond 28 days, bacterial contamination risk increases and peptide degradation accelerates — discard any remaining solution even if it appears clear. Store vials upright in the coldest part of your refrigerator, away from the door.