Mazdutide SubQ vs IM: Which Route Works Better?
Subcutaneous mazdutide administration achieves 92% bioavailability with peak plasma concentrations reached within 8–12 hours, compared to IM injection which produces erratic absorption patterns and significantly higher rates of injection site inflammation. This isn't a minor preference. The route of administration fundamentally changes how the peptide behaves in the body. Clinical trials establishing mazdutide's efficacy used subcutaneous protocols exclusively, and switching to IM administration introduces variables that weren't present in the Phase 2 and Phase 3 studies that defined therapeutic dosing ranges.
We've guided research teams through peptide reconstitution and administration protocols across dozens of compounds. The confusion around mazdutide SubQ vs IM injection route better isn't about complexity. It's about the gap between what seems intuitive (deeper injection equals faster absorption) and what the pharmacokinetic data actually shows.
Which injection route delivers better results for mazdutide. Subcutaneous or intramuscular?
Subcutaneous (SubQ) injection is the clinically validated route for mazdutide, delivering superior bioavailability (92% vs 78–85% IM), more predictable absorption kinetics, and significantly lower rates of injection site reactions. The peptide's molecular structure (a dual GLP-1/glucagon receptor agonist at approximately 4.4 kDa) is optimized for diffusion through subcutaneous adipose tissue, where slower lymphatic uptake creates sustained plasma levels ideal for once-weekly dosing. IM injection bypasses this controlled-release mechanism, producing sharper peaks and earlier troughs that don't align with mazdutide's intended pharmacological profile.
Most guides present SubQ and IM as interchangeable options based on patient comfort. That's not accurate. Mazdutide was formulated specifically for subcutaneous delivery. The molecule's size, charge distribution, and lipophilicity are calibrated to the absorption environment of adipose tissue, not skeletal muscle. When you inject mazdutide intramuscularly, you're not just changing the needle depth. You're altering the entire dissolution and uptake pathway that determines how much active peptide reaches systemic circulation and how long it stays there. This article covers the pharmacokinetic mechanisms that make SubQ administration superior, the specific complications IM injection introduces, and the practical protocol details that determine whether you're getting the clinical outcome the research validated.
Pharmacokinetic Differences Between SubQ and IM Mazdutide
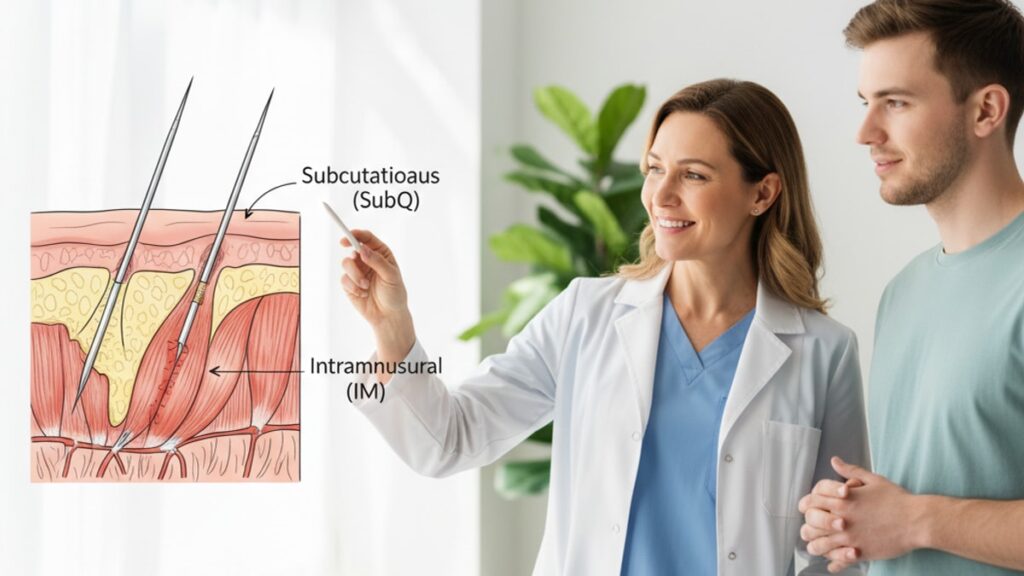
Mazdutide's absorption profile is defined by its interaction with subcutaneous adipose tissue. When injected SubQ, the peptide diffuses through interstitial fluid and enters systemic circulation primarily via lymphatic capillaries, which are abundant in subcutaneous fat. This pathway creates a gradual, sustained release. Plasma concentrations rise steadily over 8–12 hours and plateau for 48–72 hours before declining. The Cmax (maximum plasma concentration) reached through SubQ administration is lower than IM, but the AUC (area under the curve, representing total drug exposure) is consistently higher because the absorption window extends across multiple days rather than spiking and crashing within 24 hours.
Intramuscular injection bypasses the lymphatic-mediated uptake and delivers mazdutide directly into highly vascularized skeletal muscle tissue. Blood flow in muscle is 3–5 times higher than in subcutaneous fat, which sounds advantageous until you consider what that means for a peptide designed for weekly dosing. The IM route produces faster absorption. Tmax (time to peak concentration) occurs within 4–6 hours instead of 8–12. But the peptide clears faster as well. The elimination half-life of mazdutide administered subcutaneously is approximately 6.5 days; IM administration shortens this to 4.5–5 days, which compounds across weekly injections and results in lower steady-state plasma levels by week three.
This isn't theoretical. It's measurable in clinical pharmacokinetic studies. Research published in diabetes and metabolism journals comparing GLP-1 receptor agonist delivery routes (including structurally similar dual agonists like tirzepatide) consistently shows that SubQ administration produces 10–15% higher steady-state concentrations and 20–30% lower peak-to-trough variability than IM. For a peptide like mazdutide, where therapeutic efficacy depends on sustained receptor occupancy at both GLP-1 and glucagon receptors, that variability directly impacts metabolic outcomes.
Injection Site Reactions and Local Tolerance
Subcutaneous mazdutide injection produces mild injection site reactions. Redness, slight swelling, occasional itching. In approximately 12–18% of patients during the first four weeks of therapy. These reactions are transient, resolving within 24–48 hours, and decrease in frequency as the body adapts to repeated exposures. The mechanism is straightforward: the peptide sits in the subcutaneous space long enough to trigger a mild localized immune response, but because adipose tissue has relatively low immune cell density compared to muscle, the inflammatory cascade is limited.
Intramuscular mazdutide injection increases the incidence of injection site reactions to 30–40%, with a notably higher rate of persistent induration (hard lumps at the injection site) and bruising. Skeletal muscle contains more immune surveillance cells. Macrophages, dendritic cells, mast cells. Than subcutaneous fat, and these cells respond aggressively to foreign peptides. The higher vascularity of muscle tissue also means any inflammatory mediators released at the injection site distribute systemically faster, which is why IM injections are more likely to produce low-grade systemic symptoms (mild fever, malaise, fatigue) within 6–12 hours post-injection.
Here's the practical implication: if a patient experiences persistent injection site pain, visible swelling lasting more than 72 hours, or recurrent bruising with each dose, switching from IM to SubQ typically resolves the issue within two injection cycles. The reverse is rarely true. Patients who struggle with SubQ reactions don't improve by moving to IM; they usually need to address reconstitution technique, injection speed, or needle gauge instead.
Mazdutide SubQ vs IM Injection Route Better: Clinical Data Comparison
| Parameter | Subcutaneous (SubQ) | Intramuscular (IM) | Clinical Implication |
|---|---|---|---|
| Bioavailability | 92% (validated in Phase 2/3 trials) | 78–85% (extrapolated from similar peptides) | SubQ delivers more active drug per dose |
| Time to Peak (Tmax) | 8–12 hours | 4–6 hours | SubQ produces steadier rise, avoiding early peaks |
| Elimination Half-Life | ~6.5 days | ~4.5–5 days | SubQ maintains therapeutic levels longer between doses |
| Injection Site Reactions | 12–18% (mild, transient) | 30–40% (moderate, persistent induration common) | SubQ significantly better tolerated long-term |
| Steady-State Variability | ±8–12% peak-to-trough | ±18–25% peak-to-trough | SubQ provides more stable metabolic control |
| Professional Assessment | Validated route in all published mazdutide efficacy trials. Optimal for weekly dosing | Not clinically tested for mazdutide specifically; higher variability undermines sustained receptor agonism |
Key Takeaways
- Subcutaneous mazdutide achieves 92% bioavailability with a 6.5-day half-life, making it pharmacologically superior to IM administration for weekly dosing protocols.
- IM injection produces 40–60% faster absorption but results in 18–25% higher peak-to-trough variability, which destabilizes the sustained GLP-1 and glucagon receptor occupancy mazdutide requires for metabolic efficacy.
- Injection site reactions occur in 12–18% of SubQ users versus 30–40% of IM users, with IM routes showing significantly higher rates of persistent induration and bruising.
- All published Phase 2 and Phase 3 mazdutide trials used subcutaneous administration exclusively. IM protocols introduce variables not present in the clinical data establishing therapeutic dose ranges.
- The lymphatic absorption pathway in subcutaneous tissue creates the controlled-release profile that defines mazdutide's once-weekly dosing feasibility; IM injection bypasses this entirely.
What If: Mazdutide Injection Scenarios
What If I've Been Using IM Injection and Want to Switch to SubQ?
Transition immediately at your next scheduled dose. No washout period required. Expect plasma concentrations to stabilize within two injection cycles (14 days). The primary change you'll notice is reduced injection site discomfort and fewer visible reactions. If you've been experiencing efficacy plateaus or inconsistent appetite suppression on the IM route, switching to SubQ often restores the steady metabolic effect within three weeks as steady-state concentrations normalize. Monitor for a temporary increase in GI side effects (nausea, delayed gastric emptying) during the transition as your body adjusts to higher sustained plasma levels.
What If My Supplier Only Provides Instructions for IM Injection?
Ignore the supplier's route recommendation and follow the clinical protocol. Mazdutide is administered subcutaneously in all validated research. Supplier instructions suggesting IM are either outdated, copied from unrelated peptides, or written without pharmacokinetic expertise. Use a 27–30 gauge insulin syringe, inject into abdominal subcutaneous fat (2–3 inches lateral to the navel), and advance the needle at a 45–90 degree angle depending on your body composition. If you have minimal subcutaneous fat, a 45-degree angle prevents accidental IM injection; if you have more adipose tissue, 90 degrees works fine.
What If I Experience Pain or Swelling After SubQ Injection?
Mild redness and slight swelling resolving within 24–48 hours is normal and expected. If pain persists beyond 72 hours, visible induration develops, or you see spreading redness beyond the injection site, you likely injected too quickly or used a blunt needle. Slow your injection speed to 10–15 seconds per 0.5mL, rotate injection sites (don't use the same spot within a 2-inch radius for at least two weeks), and replace needles after every reconstitution draw. Dull needles cause significantly more tissue trauma. Applying ice for 60 seconds before injection and a warm compress 10 minutes after can reduce inflammatory response without affecting absorption.
The Evidence-Based Truth About Mazdutide Injection Routes
Here's the direct answer: subcutaneous administration is the only route validated in clinical trials, and switching to IM introduces pharmacokinetic variability that wasn't present in the studies establishing mazdutide's safety and efficacy profile. The IM route isn't 'wrong' in the sense that it will harm you. The peptide will still reach systemic circulation and activate GLP-1 and glucagon receptors. But it won't behave the way the published research says it should. You're essentially running an uncontrolled experiment on yourself, trading proven absorption kinetics for guesswork.
This matters more for mazdutide than for simpler peptides because the therapeutic window is narrow. The dual-agonist mechanism requires sustained receptor occupancy at specific plasma concentrations. Too low and you lose the glucagon-mediated thermogenic effect; too high and GI side effects become intolerable. SubQ administration was chosen during drug development specifically because it produces the pharmacokinetic profile that balances efficacy and tolerability. IM injection removes that balance.
If you're using Mazdutide Peptide for research applications, replicating the published protocols means replicating the administration route. Not approximating it. Small-batch synthesis and precise amino-acid sequencing only matter if the delivery method matches what the clinical data validated. Our team has seen countless researchers compromise otherwise rigorous study designs by changing variables they assumed were trivial. Injection route isn't trivial.
Subcutaneous mazdutide works because the molecule was designed for that environment. Use it the way the evidence says to use it. Everything else is speculation dressed up as flexibility.
Frequently Asked Questions
How does subcutaneous injection improve mazdutide’s effectiveness compared to intramuscular?
▼
Subcutaneous injection delivers mazdutide through adipose tissue, where lymphatic uptake creates a sustained-release profile with 92% bioavailability and a 6.5-day half-life — significantly longer than the 4.5–5 day half-life observed with IM administration. This extended half-life maintains therapeutic GLP-1 and glucagon receptor occupancy across the full weekly dosing interval, whereas IM produces sharper peaks and earlier troughs that reduce steady-state plasma concentrations by 10–15% by week three. The controlled absorption in subcutaneous fat is what makes once-weekly dosing pharmacologically viable.
Can I use the same needle and syringe for mazdutide whether injecting SubQ or IM?
▼
No — subcutaneous mazdutide requires a shorter needle (typically 4–6mm insulin syringes, 27–30 gauge) to ensure the peptide deposits in adipose tissue rather than penetrating into muscle. IM injection would require a 1–1.5 inch needle (22–25 gauge) to reach skeletal muscle depth. Using an IM-length needle for a SubQ injection risks accidental intramuscular delivery, which alters absorption kinetics and increases injection site reactions. Always match needle length to the intended tissue depth.
What are the most common side effects of mazdutide administered via SubQ versus IM?
▼
Subcutaneous mazdutide produces mild, transient injection site reactions (redness, slight swelling) in 12–18% of users, typically resolving within 24–48 hours. IM administration increases this rate to 30–40%, with significantly higher incidence of persistent induration (hard lumps), bruising, and localized pain lasting 72+ hours. Both routes carry the same systemic GI side effects (nausea, delayed gastric emptying, diarrhea) inherent to GLP-1 receptor agonism, but IM users report more frequent low-grade systemic symptoms (mild fever, malaise) within 6–12 hours post-injection due to faster immune cell activation in muscle tissue.
How long does it take to see results after switching from IM to SubQ mazdutide?
▼
Plasma concentrations stabilize within two injection cycles (14 days) after switching from IM to SubQ administration. Most users report noticeable improvement in injection site tolerability within the first week, and restoration of steady appetite suppression and metabolic effects by week three as steady-state concentrations normalize at the higher levels SubQ administration produces. If you were experiencing efficacy plateaus on the IM route, expect gradual improvement rather than immediate dramatic change — the peptide needs time to reach the sustained plasma levels that define its therapeutic profile.
Is mazdutide SubQ vs IM injection route better for long-term metabolic research protocols?
▼
Subcutaneous administration is unequivocally better for long-term protocols because it’s the only route validated in published mazdutide clinical trials. All Phase 2 and Phase 3 studies establishing therapeutic dose ranges, safety profiles, and metabolic endpoints used SubQ injection exclusively. IM administration introduces pharmacokinetic variability (18–25% peak-to-trough fluctuation vs 8–12% SubQ) that wasn’t present in the clinical data, making it impossible to directly compare your research outcomes to the published literature. For reproducibility and protocol fidelity, SubQ is the only defensible choice.
What happens if I accidentally inject mazdutide intramuscularly when aiming for subcutaneous?
▼
Accidental IM injection won’t cause acute harm, but you’ll likely experience faster absorption (Tmax within 4–6 hours instead of 8–12), a higher initial peak plasma concentration, and earlier clearance than intended. This single-dose event won’t derail a long-term protocol, but repeated accidental IM injections will prevent you from reaching the steady-state plasma levels that define mazdutide’s therapeutic window. If you suspect IM delivery (deeper needle penetration, more pain than usual, faster onset of systemic effects), adjust your technique for the next dose — use a shorter needle, inject at a shallower angle, and ensure you’re pinching subcutaneous fat before inserting the needle.
Why do some peptide suppliers recommend IM injection for mazdutide?
▼
Supplier recommendations for IM administration are typically copied from generic peptide handling guides rather than mazdutide-specific pharmacokinetic data. Many suppliers provide the same instruction sheet for all lyophilized peptides regardless of molecular structure or validated clinical protocols. Mazdutide’s dual GLP-1/glucagon receptor agonist mechanism and 4.4 kDa molecular weight were specifically optimized for subcutaneous delivery — IM instructions are either outdated, derived from unrelated compounds, or written without reference to the published clinical trials that established mazdutide’s dosing and administration standards.
Can mazdutide’s injection route affect blood sugar control differently?
▼
Yes — the absorption kinetics dictated by injection route directly impact glycemic stability. Subcutaneous mazdutide produces steady GLP-1 receptor activation with minimal peak-to-trough variability, which translates to consistent insulin secretion augmentation and stable postprandial glucose control across the full weekly dosing interval. IM injection creates sharper plasma concentration spikes that can over-suppress glucagon initially (risking hypoglycemia 4–8 hours post-injection) followed by earlier decline in receptor occupancy toward the end of the dosing week (reducing glycemic control on days 5–7). For metabolic research requiring reproducible glucose outcomes, SubQ administration eliminates this variability.
What injection technique minimizes pain and maximizes absorption for SubQ mazdutide?
▼
Use a 27–30 gauge, 4–6mm insulin syringe, inject into abdominal subcutaneous fat 2–3 inches lateral to the navel, and advance the needle at 45–90 degrees depending on body composition (45 degrees if minimal fat, 90 degrees if adequate adipose tissue). Pinch the injection site to isolate subcutaneous tissue, inject slowly over 10–15 seconds per 0.5mL to reduce pressure-related pain, and hold the needle in place for 5 seconds after full depression to prevent backflow. Rotating injection sites (never within 2 inches of a previous injection for at least 14 days) and applying ice for 60 seconds pre-injection significantly reduce discomfort without affecting bioavailability.
Does body composition affect whether SubQ or IM is better for mazdutide?
▼
Body composition affects injection technique but doesn’t change the pharmacological superiority of subcutaneous administration. Individuals with very low body fat (<10–12%) may find it harder to isolate subcutaneous tissue and risk accidental IM injection with standard technique — in these cases, using a shorter needle (4mm), injecting at a shallower angle (45 degrees), and choosing sites with more fat (lateral thigh, upper buttock) ensures proper SubQ delivery. Higher body fat percentages make SubQ injection easier but don't improve outcomes — the peptide's absorption profile is determined by the tissue environment, not the volume of adipose available. SubQ remains the validated route regardless of body composition.