FOXO4-DRI vs P21 — Mechanism, Function & Research Differences | Real Peptides
A 2017 study published in Cell by Baar et al. demonstrated that FOXO4-DRI induced apoptosis in senescent cells without affecting healthy cells. A finding that positioned it as one of the first targeted senolytics with high selectivity. The research showed that disrupting the FOXO4-p53 interaction restored p53's pro-apoptotic function specifically in senescent cells, which had been evading programmed cell death through this protein-protein binding. That mechanism is fundamentally different from what P21 does. And conflating the two reflects a surface-level misunderstanding of cellular aging biology.
We've worked with researchers examining both compounds across multiple studies. The confusion arises because both are involved in cellular senescence pathways, but their roles are opposite: FOXO4-DRI eliminates cells that have entered permanent growth arrest, while P21 enforces that arrest in the first place.
What is the difference between FOXO4-DRI and P21?
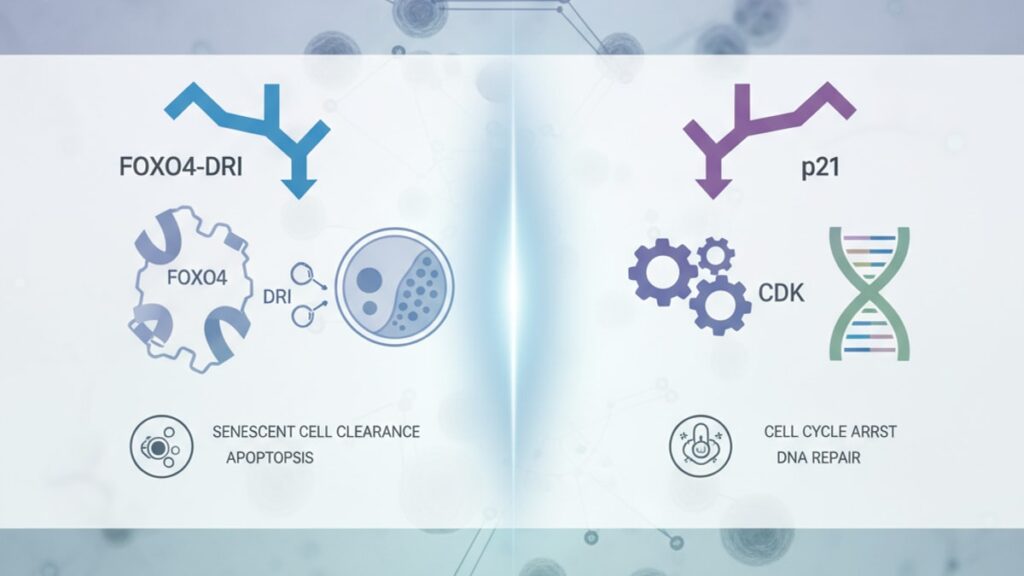
FOXO4-DRI is a synthetic peptide that disrupts the interaction between FOXO4 and p53 proteins in senescent cells, triggering apoptosis in those cells specifically. P21 is a cyclin-dependent kinase inhibitor (CDKI) that halts cell cycle progression at the G1/S checkpoint, preventing DNA-damaged or stressed cells from dividing. FOXO4-DRI acts as a senolytic. It kills senescent cells after they've formed. P21 acts as a tumor suppressor and senescence inducer. It stops cells from replicating when damage is detected, pushing them into the senescent state.
FOXO4-DRI is not a naturally occurring compound. It's a rationally designed peptide antagonist developed specifically to interfere with senescent cell survival. P21 (also called CDKN1A or WAF1/CIP1) is an endogenous protein your cells produce in response to DNA damage, oxidative stress, or oncogene activation. The difference between FOXO4-DRI and P21 is the difference between a demolition tool and a safety brake. P21 stops the car when the engine overheats; FOXO4-DRI removes the broken-down car from the road entirely. This article covers the molecular mechanisms that distinguish them, the research contexts in which each is studied, and what researchers working with either compound need to understand about their divergent biological roles.
The Core Biological Distinction: Senolytic vs Anti-Proliferative
P21 is a cyclin-dependent kinase inhibitor encoded by the CDKN1A gene. When cells experience genotoxic stress. UV radiation, oxidative damage, telomere shortening. P53 (the 'guardian of the genome') transcriptionally upregulates P21 expression. P21 then binds to cyclin-CDK complexes (specifically cyclin E-CDK2 and cyclin D-CDK4/6), preventing phosphorylation of the retinoblastoma protein (Rb). Unphosphorylated Rb sequesters E2F transcription factors, blocking entry into S phase and halting DNA replication. This is the G1/S checkpoint arrest. The cell stops dividing but remains metabolically active.
FOXO4-DRI operates at the opposite end of the senescence timeline. Once cells have entered irreversible growth arrest and adopted the senescent phenotype, they resist apoptosis through several mechanisms. One of which is the FOXO4-p53 interaction. In senescent cells, FOXO4 binds to p53 and sequesters it in the nucleus, preventing p53 from activating pro-apoptotic genes like PUMA and NOXA. FOXO4-DRI is a modified peptide derived from the p53-binding domain of FOXO4. It competes for the same binding site, displacing endogenous FOXO4 and liberating p53 to trigger intrinsic apoptosis. The Baar study showed this effect was selective: in non-senescent cells, p53 isn't sequestered by FOXO4 in the same way, so FOXO4-DRI has minimal impact.
Our team has reviewed this mechanism across multiple cellular aging models. The selectivity comes from the fact that FOXO4-p53 binding is dramatically upregulated in senescent cells. So FOXO4-DRI preferentially disrupts a survival pathway that only senescent cells rely on.
FOXO4-DRI vs P21: Mechanism, Function & Research — Comparison
The table below contrasts FOXO4-DRI and P21 across molecular function, biological role, research application, and mechanism of action.
| Feature | FOXO4-DRI | P21 (CDKN1A) | Professional Assessment |
|---|---|---|---|
| Molecular Classification | Synthetic peptide antagonist (modified FOXO4 fragment) | Endogenous cyclin-dependent kinase inhibitor (CDKI) | FOXO4-DRI is exogenous and rationally designed; P21 is a naturally expressed protein |
| Primary Mechanism | Disrupts FOXO4-p53 interaction in senescent cells, liberating p53 to activate apoptotic pathways | Inhibits cyclin-CDK2/4/6 complexes, preventing Rb phosphorylation and blocking G1/S transition | FOXO4-DRI acts post-senescence; P21 acts during senescence induction |
| Biological Role | Senolytic. Induces apoptosis in senescent cells | Tumor suppressor and senescence inducer. Halts cell cycle in response to stress | Opposite ends of the senescence spectrum: enforcement vs elimination |
| Selectivity | Preferentially affects senescent cells (low toxicity to proliferating cells) | Broadly expressed in stress conditions; affects both normal and stressed cells | FOXO4-DRI's selectivity is its defining feature; P21 is context-dependent |
| Research Context | Anti-aging studies, senolytic therapy, tissue rejuvenation models | Cancer biology, DNA damage response, cellular stress pathways | FOXO4-DRI is niche and experimental; P21 is foundational in cell cycle research |
| Clinical Translation Stage (2026) | Preclinical. No FDA-approved human therapies exist | Therapeutic target in oncology (CDK4/6 inhibitors modulate P21 downstream) | FOXO4-DRI remains research-grade; P21 pathway is clinically validated in cancer treatment |
Key Takeaways
- FOXO4-DRI is a synthetic senolytic peptide that selectively induces apoptosis in senescent cells by disrupting the FOXO4-p53 survival interaction.
- P21 is an endogenous cyclin-dependent kinase inhibitor that halts the cell cycle at the G1/S checkpoint in response to DNA damage or stress, inducing senescence rather than eliminating it.
- The 2017 Baar et al. Cell study demonstrated FOXO4-DRI restored fur density and renal function in aged mice. Evidence of functional tissue rejuvenation through senescent cell clearance.
- P21 is transcriptionally activated by p53 and functions as a tumor suppressor by preventing damaged cells from replicating, but it does not kill those cells.
- FOXO4-DRI's selectivity for senescent cells arises because the FOXO4-p53 interaction is upregulated specifically in senescent phenotypes. Healthy proliferating cells are largely unaffected.
- As of 2026, FOXO4-DRI remains a research-grade compound with no FDA-approved therapeutic applications, while P21 pathway modulation is clinically established in oncology via CDK4/6 inhibitors.
What If: FOXO4-DRI and P21 Scenarios
What If You're Designing a Senolytic Protocol and Considering P21 Inhibition?
Don't conflate P21 inhibition with senolytic activity. Inhibiting P21 would prevent cells from entering senescence in the first place, not clear existing senescent cells. If you block P21 function in already-senescent cells, you don't reverse their phenotype; you may impair their ability to maintain growth arrest, potentially allowing damaged cells to re-enter the cycle. The senolytic approach targets cells that are already senescent and resistant to apoptosis. FOXO4-DRI does this by restoring p53 apoptotic signaling. P21 operates upstream of that state.
What If You're Using P21 as a Senescence Marker and See FOXO4-DRI Studies Referenced?
P21 upregulation is a canonical marker of cellular senescence (along with SA-β-gal, p16^INK4a^, and SASP cytokines). FOXO4-DRI studies use P21 expression to identify senescent cells before treatment. The peptide is then applied to eliminate those P21-positive cells. The presence of P21 doesn't mean the cell will respond to FOXO4-DRI; the response depends on whether FOXO4-p53 binding is active. In some senescent cell types, alternative survival pathways (BCL-2 family proteins, for example) dominate, and FOXO4-DRI may be less effective.
What If You're Researching Tumor Suppression and Encounter FOXO4-DRI Literature?
P21's role as a tumor suppressor is well-established. It prevents cells with oncogenic mutations or DNA damage from proliferating. FOXO4-DRI does not replicate this function. In fact, eliminating senescent cells (which are often pre-malignant cells held in permanent arrest) raises theoretical cancer risk if done non-selectively or in contexts where senescence is acting as a tumor suppressor barrier. The Baar study addressed this by demonstrating selectivity for senescent over proliferating cells, but the long-term oncogenic risk of chronic senolytic use remains an open question in 2026.
The Blunt Truth About FOXO4-DRI and P21
Here's the honest answer: if you're treating these compounds as functionally similar, you've misunderstood cellular senescence biology at a foundational level. P21 is the handbrake. It stops the cell when damage is detected and holds it in place. FOXO4-DRI is the controlled demolition. It removes the stopped cell from the system entirely. Researchers conflate them because both appear in senescence literature, but their mechanisms are opposite. P21 creates senescent cells; FOXO4-DRI destroys them. One is a protective response; the other is an interventional tool. The difference between FOXO4-DRI and P21 is the difference between prevention and elimination. Mixing them up in experimental design or literature interpretation is a fundamental error.
Why the FOXO4-p53 Interaction Matters More in Senescent Cells
In proliferating cells, p53 activity is tightly regulated. It's kept at low levels under normal conditions and rapidly activated in response to acute stress. When DNA damage occurs, p53 stabilizes, translocates to the nucleus, and either triggers repair (via P21-mediated arrest) or apoptosis (via PUMA, NOXA, BAX). The decision between arrest and death depends on the severity and context of the damage.
In senescent cells, this regulatory balance shifts. Chronic p53 activation would normally trigger apoptosis, but senescent cells evade this through multiple mechanisms. One key mechanism is the upregulation of FOXO4, which binds to p53 in the nucleus and prevents it from activating pro-apoptotic target genes. This allows senescent cells to maintain high p53 levels (which drives the senescent phenotype through P21 and other effectors) without dying. The 2017 Baar study exploited this: by designing a peptide that disrupts FOXO4-p53 binding, they restored p53's apoptotic function specifically in cells where this interaction was pathologically elevated.
Our experience with P21 and related cell cycle research tools shows that understanding these regulatory shifts is critical for interpreting experimental outcomes. P21 expression tells you a cell has activated the senescence program. It doesn't tell you whether that cell will survive long-term or be cleared. FOXO4-DRI studies assume the cells are already P21-positive and senescent; the peptide's role is clearance, not induction.
The pharmacological challenge with FOXO4-DRI is delivery and selectivity at the organismal level. In vitro, the peptide shows high selectivity for senescent versus proliferating cells. In vivo, achieving therapeutic concentrations in target tissues (liver, kidney, adipose) without off-target effects in rapidly dividing tissues (gut epithelium, bone marrow) is harder. The Baar study used fast-acting chemotherapy to induce acute senescence in mice, then administered FOXO4-DRI. This proof-of-concept showed efficacy but doesn't address chronic low-level senescent cell accumulation in natural aging. As of 2026, no published human trials have tested FOXO4-DRI systemically, and the compound remains research-grade. Real Peptides offers high-purity research tools like Thymalin and other peptides for cellular aging studies. Compounds synthesized with exact amino-acid sequencing for lab reliability.
P21, by contrast, is not used as an exogenous therapeutic in the same way FOXO4-DRI might be. Instead, P21 pathway modulation happens through upstream interventions: CDK4/6 inhibitors (palbociclib, ribociclib, abemaciclib) are FDA-approved cancer therapies that work by mimicking P21's cell cycle arrest effect. These drugs don't directly inhibit P21. They inhibit the kinases P21 would normally block, achieving the same G1/S arrest. This distinction matters: you can't 'administer P21' the way you administer FOXO4-DRI; P21 is a downstream effector of endogenous stress responses.
The difference between FOXO4-DRI and P21 is ultimately about intervention level. FOXO4-DRI is a targeted exogenous tool designed to correct a specific survival defect in senescent cells. P21 is an endogenous checkpoint protein that cells produce in response to damage. It's not a drug, it's a biological readout. Modulating the pathways that regulate P21 (p53, CDKs, Rb) is how therapeutic intervention happens, not by delivering P21 itself. For researchers designing aging or cancer studies, this distinction determines whether you're measuring a response, inducing a state, or eliminating a population. Three entirely different experimental aims that require different compounds and different endpoints.
FOXO4-DRI's role in the broader senolytic landscape positions it alongside compounds like dasatinib + quercetin (D+Q), navitoclax (ABT-263), and fisetin. All of which target senescent cell survival pathways but through different mechanisms. D+Q works by inhibiting pro-survival kinases and BCL-2 family proteins; navitoclax directly inhibits BCL-2, BCL-xL, and BCL-w; fisetin modulates multiple senescence-associated pathways including PI3K/AKT. FOXO4-DRI's specificity for the FOXO4-p53 axis makes it complementary rather than redundant. Some senescent cell types depend more on BCL-2, others on FOXO4-p53, and still others on alternative pathways. Combination senolytic strategies are under investigation in 2026 to address this heterogeneity. If your research involves senescent cell clearance, understanding which survival pathway dominates in your model system determines which senolytic is appropriate. And P21 expression alone doesn't answer that question.
Frequently Asked Questions
What is the primary difference between FOXO4-DRI and P21 in terms of cellular function?
▼
FOXO4-DRI is a synthetic senolytic peptide that induces apoptosis in senescent cells by disrupting the FOXO4-p53 survival interaction, while P21 is an endogenous cyclin-dependent kinase inhibitor that halts the cell cycle at the G1/S checkpoint in response to stress or DNA damage. FOXO4-DRI eliminates cells that have already entered senescence; P21 enforces the senescent state in the first place. They operate at opposite ends of the senescence timeline — P21 is the brake, FOXO4-DRI is the demolition.
Can FOXO4-DRI be used to prevent cells from becoming senescent?
▼
No — FOXO4-DRI does not prevent senescence induction. It acts on cells that are already senescent and evading apoptosis through the FOXO4-p53 interaction. Preventing senescence would require modulating upstream pathways like P21, p53 activation, or DNA damage response mechanisms. FOXO4-DRI’s role is clearance of existing senescent cells, not prevention of their formation.
Is P21 a senolytic compound like FOXO4-DRI?
▼
No — P21 is not a senolytic. It is a tumor suppressor and cell cycle checkpoint protein that induces senescence by halting cell division when damage is detected. Senolytics like FOXO4-DRI work by killing senescent cells after they have formed. Inhibiting P21 would not clear senescent cells; it would prevent cells from entering senescence in the first place, which could allow damaged cells to continue dividing.
What did the 2017 Baar study demonstrate about FOXO4-DRI efficacy?
▼
The 2017 Baar et al. study published in *Cell* demonstrated that FOXO4-DRI selectively induced apoptosis in senescent cells by disrupting the FOXO4-p53 interaction, restoring p53’s pro-apoptotic function. In aged mice, FOXO4-DRI treatment restored fur density, improved renal function, and extended healthspan markers — evidence of functional tissue rejuvenation through senescent cell clearance. The study also showed minimal toxicity to non-senescent cells, confirming selectivity.
How does P21 contribute to tumor suppression?
▼
P21 prevents cells with DNA damage or oncogenic mutations from replicating by inhibiting cyclin-CDK complexes, which blocks cell cycle progression at the G1/S checkpoint. This gives the cell time to repair damage or enter permanent senescence rather than dividing with genomic instability. P21 is transcriptionally activated by p53 in response to genotoxic stress and is considered one of the canonical tumor suppressor pathways — loss of P21 function is associated with increased cancer risk.
Can FOXO4-DRI affect healthy proliferating cells?
▼
FOXO4-DRI shows minimal toxicity to healthy proliferating cells because the FOXO4-p53 interaction it disrupts is upregulated specifically in senescent cells. In non-senescent cells, p53 is not sequestered by FOXO4 in the same manner, so FOXO4-DRI binding has little functional effect. The Baar study confirmed this selectivity in vitro and in vivo, though off-target effects at very high doses or in specific tissue contexts remain under investigation as of 2026.
Why is P21 used as a senescence marker if it doesn’t eliminate senescent cells?
▼
P21 is upregulated when cells enter senescence in response to DNA damage, oxidative stress, or oncogene activation, making it a reliable biomarker for identifying senescent cells in research models. Its presence indicates a cell has activated the senescence program, even though P21 itself does not kill the cell. Researchers use P21 expression (alongside SA-β-gal, p16, and SASP markers) to identify senescent populations before applying senolytics like FOXO4-DRI to clear them.
What is the FOXO4-p53 interaction and why does it matter for senescent cell survival?
▼
In senescent cells, FOXO4 binds to p53 in the nucleus and prevents p53 from activating pro-apoptotic genes like *PUMA* and *NOXA*. This allows senescent cells to maintain high p53 levels (driving the senescent phenotype) without triggering apoptosis. FOXO4-DRI disrupts this interaction, liberating p53 to activate apoptotic pathways and induce cell death specifically in senescent cells where this survival mechanism is active.
Are there FDA-approved therapies using FOXO4-DRI as of 2026?
▼
No — FOXO4-DRI remains a research-grade compound with no FDA-approved human therapeutic applications as of 2026. It is used in preclinical aging and senescence studies but has not progressed to clinical trials for systemic senolytic therapy. All current FOXO4-DRI research is conducted in vitro or in animal models.
How do CDK4/6 inhibitors relate to P21 function?
▼
CDK4/6 inhibitors (palbociclib, ribociclib, abemaciclib) are FDA-approved cancer drugs that mimic P21’s cell cycle arrest effect by directly inhibiting cyclin-dependent kinases 4 and 6. P21 normally inhibits these same kinases in response to p53 activation — CDK4/6 inhibitors achieve the same G1/S arrest pharmacologically without requiring P21 expression. This is how the P21 pathway is therapeutically modulated in oncology.
What other senolytics are FOXO4-DRI compared to in research?
▼
FOXO4-DRI is compared to dasatinib + quercetin (D+Q), navitoclax (ABT-263), and fisetin — all of which target senescent cell survival pathways but through different mechanisms. D+Q inhibits pro-survival kinases and BCL-2 family proteins; navitoclax directly inhibits BCL-2, BCL-xL, and BCL-w; FOXO4-DRI specifically disrupts the FOXO4-p53 interaction. Senescent cell heterogeneity means different cell types depend on different survival pathways, making combination senolytic strategies an active area of investigation in 2026.
If I inhibit P21, will that clear senescent cells from tissue?
▼
No — inhibiting P21 would not clear existing senescent cells. P21 inhibition prevents cells from entering senescence by allowing them to bypass the G1/S checkpoint despite DNA damage, which could permit damaged cells to continue dividing. Clearing senescent cells requires senolytics like FOXO4-DRI that target survival pathways active in already-senescent populations, not the pathways that induce senescence.