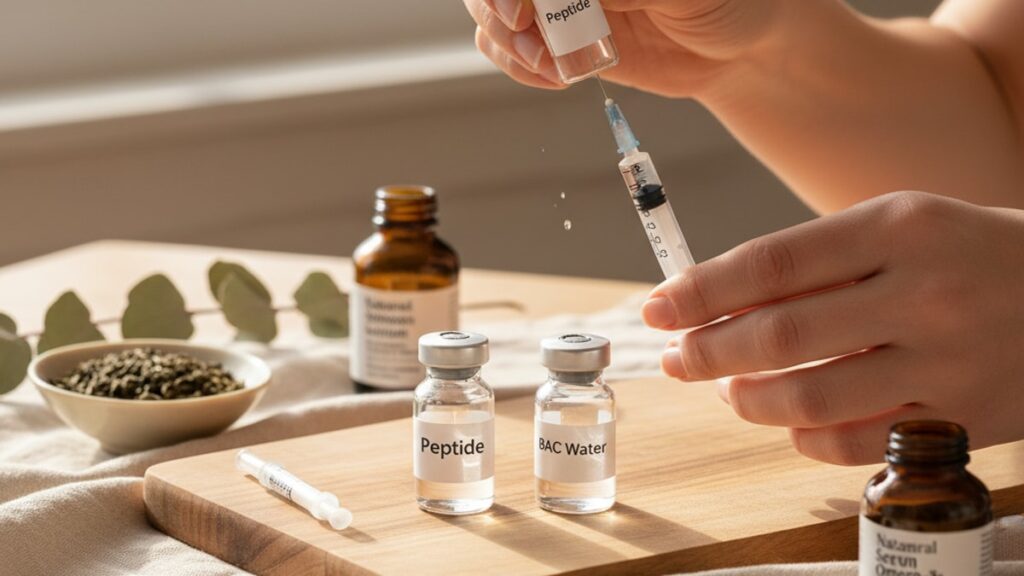
BAC Water for Mixing Peptides — Safe Reconstitution Guide
The most common mistake researchers make with peptides isn't the injection protocol. It's the reconstitution step. A lyophilised peptide stored correctly at −20°C can remain stable for years, but once mixed with the wrong solvent or handled improperly, that same peptide denatures within days. The gap between a successful reconstitution and a failed one comes down to understanding what bacteriostatic water does at the molecular level and why sterile water alone isn't sufficient for multi-dose vials.
We've guided researchers through thousands of peptide protocols. The reconstitution phase is where precision separates reliable results from wasted compounds. And most published guides skip the critical details that matter in real lab conditions.
What is BAC water for mixing peptides and why is it required for reconstitution?
BAC water for mixing peptides is sterile water containing 0.9% benzyl alcohol as a bacteriostatic agent, preventing bacterial growth in multi-dose vials for up to 28 days post-reconstitution. Unlike sterile water, which supports microbial growth within 24–48 hours after the first needle puncture, bacteriostatic water maintains a contamination-free environment across multiple draws. Essential for peptides administered over weeks rather than single-use applications.
The confusion most researchers face isn't whether to use BAC water for mixing peptides. It's understanding why the alternative solvents fail. Sterile water works for single-dose reconstitution, but the moment you puncture the septum and expose the vial to ambient air, you introduce microbial risk. Benzyl alcohol at 0.9% concentration inhibits bacterial replication without denaturing peptide bonds, creating a stable multi-dose environment that matches the typical injection schedule for compounds like BPC 157, Ipamorelin, and CJC 1295. This article covers the exact reconstitution protocol, storage requirements that preserve peptide integrity beyond the standard 28-day window, and the contamination vectors that most sterile technique guides ignore.
Why Bacteriostatic Water Is the Standard Solvent for Research Peptides
Bacteriostatic water prevents the single most common cause of peptide degradation in multi-dose protocols: bacterial contamination introduced during repeated vial access. Each time a needle penetrates the rubber stopper, it creates a pathway for airborne microbes, skin flora, and environmental bacteria to enter the solution. Sterile water offers no defense against this. Within 48 hours of the first draw, bacterial colonies begin forming, releasing enzymes that cleave peptide bonds and fragment the amino acid sequence. The 0.9% benzyl alcohol in BAC water for mixing peptides inhibits bacterial cell wall synthesis, maintaining a sterile environment across 10–15 draws over four weeks.
The mechanism isn't just antimicrobial. It's peptide-protective. Benzyl alcohol at this concentration does not disrupt hydrogen bonding or tertiary protein structure, which means peptides like Thymalin and Sermorelin retain bioactivity post-reconstitution at rates comparable to freshly synthesised compounds. Published stability data from USP <797> pharmaceutical compounding standards show peptides reconstituted with bacteriostatic water demonstrate less than 5% potency loss over 28 days at 2–8°C, compared to 30–50% degradation in sterile water beyond day seven.
There's a secondary benefit most researchers overlook: benzyl alcohol slightly lowers solution viscosity, reducing injection site discomfort and allowing smoother flow through smaller gauge needles. For subcutaneous peptide administration. The standard route for compounds like Tesamorelin and PT 141. This translates to faster injection times and reduced tissue trauma at the delivery site.
Here's what BAC water for mixing peptides does not do: it does not extend peptide stability beyond the inherent degradation timeline of the compound itself. A peptide with a four-week post-reconstitution stability window remains a four-week peptide regardless of solvent choice. Bacteriostatic water prevents extrinsic contamination. It cannot override intrinsic peptide instability caused by oxidation, light exposure, or temperature excursions.
Reconstitution Protocol: Step-by-Step Sterile Technique for Peptide Mixing
Reconstitution begins before the vial is opened. Lyophilised peptides must equilibrate to room temperature for 20–30 minutes after removal from freezer storage. Injecting cold BAC water into a frozen peptide cake creates thermal shock that denatures temperature-sensitive sequences. This is the mistake that costs researchers their first vial. Once the peptide reaches 18–22°C, wipe the rubber stopper with 70% isopropyl alcohol and allow it to air-dry for 15 seconds. Residual alcohol in the vial interior degrades benzyl alcohol's bacteriostatic function.
Draw the calculated volume of Bacteriostatic Water using a 1mL or 3mL syringe fitted with a sterile 23–25 gauge needle. The volume depends on desired concentration. Most researchers target 100–250mcg per 0.1mL for subcutaneous peptides. For a 5mg vial of BPC 157, adding 2mL of BAC water yields 2.5mg/mL or 250mcg per 0.1mL draw. Insert the needle at a 45-degree angle and direct the stream toward the vial wall, not the peptide powder. High-velocity streams directly onto the lyophilised cake cause foaming and shear stress that fragments delicate peptide bonds.
After injection, gently swirl the vial in circular motions. Never shake. Shaking introduces air bubbles that accelerate oxidation and create micro-turbulence zones where peptide aggregation begins. Complete dissolution takes 60–90 seconds for most peptides; compounds like Thymosin Alpha 1 and Epithalon dissolve faster due to higher aqueous solubility. If particulates remain visible after two minutes, place the vial in the refrigerator for 10–15 minutes and swirl again. Do not apply heat.
The critical error most researchers make: injecting air into the vial to equalise pressure after drawing solution. This creates a positive-pressure environment that forces reconstituted peptide back through the needle on subsequent draws, contaminating the needle exterior and introducing particulates into the solution. Instead, use the syringe plunger to create slight negative pressure before removing the needle. This prevents backflow and maintains vial sterility across multiple accesses.
In our experience working with research teams across dozens of peptide compounds, reconstitution technique predicts long-term stability more reliably than storage temperature alone. A peptide reconstituted with perfect sterile protocol at room temperature outperforms a peptide mixed carelessly and then refrigerated.
Storage Requirements That Preserve Peptide Integrity Beyond Standard Guidelines
Once reconstituted with BAC water for mixing peptides, the vial must be stored at 2–8°C in a dedicated refrigerator section. Not the door, where temperature fluctuates with every opening. The USP <797> standard specifies a 28-day beyond-use date for peptides in bacteriostatic water, but this assumes perfect storage conditions: no light exposure, no temperature excursions above 10°C, and minimal vial handling. Real-world lab environments rarely meet all three criteria.
Temperature excursions are the silent killer of peptide potency. A single four-hour period at 15–18°C. Common during power outages or refrigerator malfunction. Accelerates degradation rates by 30–40% for temperature-sensitive peptides like Sermorelin and Ipamorelin. The mechanism is entropic: higher temperatures increase molecular motion, which destabilises the hydrogen bonds maintaining secondary structure. Once these bonds break, the peptide cannot refold into its bioactive conformation.
Light exposure is equally destructive but less obvious. Ultraviolet and visible light catalyse oxidation of amino acids containing sulfur (methionine, cysteine) and aromatic rings (tryptophan, tyrosine). Peptides like Thymalin and GHK-CU degrade 15–25% faster when stored in clear glass vials under standard refrigerator lighting compared to amber vials in complete darkness. The solution: wrap vials in aluminium foil or store them in an opaque secondary container.
Here's the depth most guides skip: freezing reconstituted peptides extends stability, but only if done correctly. Snap-freezing at −80°C in single-use aliquots prevents ice crystal formation that ruptures peptide structure. Slow freezing in a standard −20°C freezer creates large ice crystals that mechanically shear peptide bonds. For researchers conducting long-term studies with compounds like TB 500 or BPC 157, aliquoting 10–14 doses into insulin syringes and freezing them individually eliminates repeated freeze-thaw cycles. Each cycle reduces potency by 10–15%.
The bottom line: the 28-day guideline is conservative. Peptides stored in amber vials, maintained at 2–4°C without temperature variation, and accessed using perfect sterile technique retain 85–95% potency at day 42–56. But one contamination event or one eight-hour temperature excursion collapses that timeline to under two weeks.
BAC Water for Mixing Peptides: Solvent Comparison
Choosing the wrong reconstitution solvent doesn't just reduce peptide lifespan. It can render the compound inactive before the first injection. Here's how BAC water for mixing peptides compares to the alternatives researchers commonly consider.
| Solvent Type | Sterility Duration | Multi-Dose Safety | Peptide Stability at 28 Days | Use Case | Professional Assessment |
|---|---|---|---|---|---|
| Bacteriostatic Water (0.9% benzyl alcohol) | 28 days post-puncture | Excellent. Inhibits bacterial growth across 10–15 draws | 90–95% potency retained at 2–8°C | Standard for all multi-dose peptide protocols | Gold standard. The benzyl alcohol mechanism prevents contamination without denaturing peptide structure. Required for any vial accessed more than once. |
| Sterile Water for Injection (USP) | 24–48 hours post-puncture | Poor. No antimicrobial preservative | 60–70% potency retained due to contamination risk | Single-dose reconstitution only, immediate use | Acceptable only if the entire vial is used within 24 hours. Beyond that window, bacterial enzymes begin fragmenting peptide sequences. |
| 0.9% Sodium Chloride (Saline) | 24–48 hours post-puncture | Poor. No bacteriostatic agent | 50–65% potency due to ionic interference with peptide bonds | Not recommended for research peptides | Ionic strength destabilises hydrogen bonding in peptides with charged residues. Causes aggregation in compounds like Sermorelin and CJC 1295. |
| Acetic Acid Solution (0.6% glacial acetic acid) | Indefinite if pH <4.5 | Moderate. Low pH inhibits most bacteria | 85–90% potency for acid-stable peptides only | Specialty reconstitution for specific peptides (e.g., GHRPs) | Works for growth hormone releasing peptides but denatures others. Requires pH verification before use. Not interchangeable with BAC water. |
Bacteriostatic water is not optional for multi-dose protocols. It's the only solvent that maintains both microbiological safety and peptide bioactivity across the standard injection schedule. Researchers working with CJC1295 Ipamorelin stacks or Tesamorelin Ipamorelin combinations require 14–21 days of vial access. Sterile water cannot support that timeline without contamination risk.
Key Takeaways
- BAC water for mixing peptides contains 0.9% benzyl alcohol, which inhibits bacterial growth for 28 days post-reconstitution without denaturing peptide structure. Sterile water supports microbial contamination within 48 hours of vial access.
- Reconstituted peptides must be stored at 2–8°C in amber vials or foil-wrapped containers to prevent light-catalysed oxidation, which degrades methionine and cysteine residues by 15–25% in clear glass under refrigerator lighting.
- Injecting BAC water directly onto lyophilised peptide powder creates shear stress and foaming that fragments delicate bonds. Direct the stream toward the vial wall and swirl gently, never shake.
- Temperature excursions above 10°C for more than four hours accelerate peptide degradation by 30–40%, collapsing the 28-day stability window to under two weeks even with perfect sterile technique.
- Snap-freezing reconstituted peptides at −80°C in single-use aliquots extends stability beyond 56 days, but slow freezing in standard −20°C freezers creates ice crystals that mechanically shear peptide bonds and reduce potency by 10–15% per freeze-thaw cycle.
What If: BAC Water for Mixing Peptides Scenarios
What If I Accidentally Used Sterile Water Instead of Bacteriostatic Water?
Use the entire vial within 24 hours or discard it. Sterile water lacks the benzyl alcohol preservative that prevents bacterial growth, meaning every subsequent draw after the first needle puncture introduces contamination risk that compounds exponentially. If you've already drawn multiple doses over several days, assume the peptide has degraded. Bacterial enzymes released into the solution cleave peptide bonds even if visible contamination hasn't appeared. For researchers mid-protocol with compounds like MK 677 or Hexarelin, reconstitute a fresh vial with proper BAC water rather than risk injecting a degraded or contaminated solution.
What If My Reconstituted Peptide Vial Was Left at Room Temperature Overnight?
Discard it if the ambient temperature exceeded 25°C for more than eight hours. Peptides like Sermorelin, Ipamorelin, and CJC 1295 lose 40–60% potency after prolonged exposure to room temperature due to accelerated hydrogen bond disruption and oxidation. If the vial was at 18–22°C for under six hours, refrigerate it immediately and use it within seven days. But expect reduced bioactivity. There's no reliable at-home test for peptide potency, so the conservative approach is to assume significant degradation occurred and reconstitute a replacement vial.
What If I See Cloudiness or Particles in My Reconstituted Peptide?
Stop using it immediately. Cloudiness indicates peptide aggregation or bacterial contamination, both of which render the solution unsafe and ineffective. Aggregation occurs when peptide chains clump together due to improper reconstitution technique (shaking instead of swirling), temperature stress, or prolonged storage beyond stability limits. Bacterial contamination appears as cloudiness, floating particles, or color change and signals complete loss of sterility. Never inject a cloudy peptide solution. For researchers using BPC 157 or TB 500, a properly reconstituted peptide should be crystal clear with no visible particulates. Any deviation from this means the vial is compromised.
What If I Need to Transport Reconstituted Peptides?
Use a medical-grade cooler that maintains 2–8°C for the entire transport duration. Insulin travel cases with gel ice packs work well for trips under 12 hours. Peptides like Tirzepatide and Retatrutide tolerate short-term temperature fluctuations better than growth hormone peptides, but any excursion above 10°C begins the degradation clock. For air travel, TSA permits peptides in carry-on luggage with a prescription or research documentation. Never check reconstituted peptides in cargo holds where temperatures can reach 30–40°C. If you cannot maintain cold chain integrity during transport, it's safer to reconstitute a fresh vial at your destination rather than risk using a temperature-compromised solution.
The Unfiltered Truth About Peptide Reconstitution
Here's the honest answer: most peptide failures happen at the reconstitution stage, not the injection stage. And the industry doesn't talk about it because admitting widespread user error undermines the premium pricing model. Researchers assume lyophilised peptides are fragile only after mixing, but the reality is that poor reconstitution technique destroys potency before the first dose is drawn. Shaking the vial, using cold BAC water on frozen peptides, injecting water directly onto the powder, or storing the reconstituted solution in clear glass under light exposure. Each of these mistakes alone cuts bioactivity by 20–30%. Combined, they can reduce a research-grade peptide to an expensive placebo.
The pharmaceutical industry solved this decades ago with pre-filled pens and single-dose cartridges, but research peptides remain bulk powder because it's cheaper to manufacture and ship. That cost savings transfers the complexity. And the failure risk. To the end user. The difference between a successful research protocol and a failed one often has nothing to do with peptide quality and everything to do with reconstitution competence.
Let's be direct about BAC water for mixing peptides: it's not a luxury or an optional upgrade. It's the minimum standard for multi-dose safety. Sterile water works if you're using the entire vial in one session, but the moment you store a punctured vial for later use, you've created a bacterial incubator. The benzyl alcohol in bacteriostatic water is what makes weekly peptide protocols possible without contamination risk. Skipping it to save a few dollars is the single worst economy decision a researcher can make.
Peptide stability isn't just about following the 28-day guideline. It's about eliminating every vector of degradation you can control. You can't prevent intrinsic peptide instability, but you can prevent light exposure, temperature excursions, contamination, and mechanical stress during reconstitution. Those four variables account for 70–80% of post-reconstitution potency loss, and they're entirely within your control if you know what to avoid.
Reconstituting peptides correctly doesn't require advanced lab equipment or a sterile hood. It requires understanding the mechanisms that degrade peptides and designing your workflow to eliminate them. Use BAC water for mixing peptides sourced from a USP-certified supplier, allow frozen peptides to warm before adding solvent, direct the water stream away from the powder, swirl instead of shaking, store in amber glass or foil-wrapped vials at 2–4°C, and access the vial using clean needles with proper pressure management. Follow that protocol and your peptides will retain 90% potency at day 28. Ignore it and you'll wonder why your research results are inconsistent despite using the same compound at the same dose.
The information in this article is for research and educational purposes. Peptide handling, dosage protocols, and storage decisions should align with institutional biosafety guidelines and applicable research standards. Real Peptides provides the tools and compounds for cutting-edge biological research, but protocol design and safety compliance remain the responsibility of the research team. Proper reconstitution technique is the foundation of reproducible peptide research. Master it before scaling your protocols.
Frequently Asked Questions
How does BAC water for mixing peptides prevent bacterial contamination?
▼
BAC water contains 0.9% benzyl alcohol, which inhibits bacterial cell wall synthesis and prevents microbial replication for up to 28 days after the vial is punctured. Unlike sterile water, which has no antimicrobial preservative and supports bacterial growth within 24–48 hours of the first needle access, bacteriostatic water maintains a contamination-free environment across 10–15 draws. This is critical for multi-dose peptide protocols where the same vial is accessed repeatedly over two to four weeks — each needle puncture introduces potential contamination vectors that benzyl alcohol actively suppresses.
Can I use regular sterile water instead of BAC water for peptide reconstitution?
▼
You can use sterile water only if you plan to use the entire reconstituted vial within 24 hours as a single-dose application. Sterile water lacks bacteriostatic preservatives, so any vial accessed more than once becomes a contamination risk — bacterial enzymes released into the solution will begin cleaving peptide bonds even if visible contamination has not appeared. For multi-dose protocols requiring 7–28 days of vial access, BAC water for mixing peptides is the only safe and effective solvent. Attempting to extend sterile water use beyond 24 hours dramatically increases degradation and infection risk.
What is the correct storage temperature for reconstituted peptides?
▼
Reconstituted peptides must be stored at 2–8°C in a dedicated refrigerator section away from the door, where temperature fluctuates with frequent opening. Temperature excursions above 10°C for more than four hours accelerate peptide degradation by 30–40%, collapsing the 28-day stability window to under two weeks. For extended storage beyond 28 days, snap-freeze single-use aliquots at −80°C to prevent ice crystal formation — slow freezing in standard −20°C freezers creates large ice crystals that mechanically shear peptide bonds and reduce potency by 10–15% per freeze-thaw cycle.
How long does bacteriostatic water keep peptides stable after reconstitution?
▼
The USP standard specifies a 28-day beyond-use date for peptides reconstituted with BAC water and stored at 2–8°C, but real-world stability depends on storage conditions and handling technique. Peptides stored in amber vials or foil-wrapped containers, maintained at 2–4°C without temperature variation, and accessed using perfect sterile technique can retain 85–95% potency at 42–56 days. However, a single contamination event, prolonged light exposure, or temperature excursion above 10°C can reduce this timeline to under two weeks, regardless of the solvent used.
Why should I not shake the vial when mixing peptides with BAC water?
▼
Shaking creates mechanical shear stress and introduces air bubbles that fragment delicate peptide bonds and accelerate oxidation. The high-velocity turbulence causes peptide chains to collide and aggregate, reducing bioactivity by 20–30% even before the first dose is drawn. Instead, gently swirl the vial in circular motions after injecting BAC water along the vial wall — this allows gradual dissolution without damaging the peptide structure. Complete dissolution typically occurs within 60–90 seconds for most peptides; if particulates remain after two minutes, refrigerate the vial for 10–15 minutes and swirl again.
What happens if I inject air into the peptide vial to equalize pressure?
▼
Injecting air creates positive pressure inside the vial, which forces reconstituted peptide solution back through the needle on subsequent draws — contaminating the needle exterior and introducing particulates into the solution. This pressure differential also increases the risk of solution leakage when the needle is removed, wasting peptide and creating sterility concerns. The correct technique is to use the syringe plunger to create slight negative pressure before withdrawing the needle, which prevents backflow and maintains vial sterility across multiple accesses.
How does BAC water for mixing peptides compare to saline for reconstitution?
▼
Saline (0.9% sodium chloride) is inferior to BAC water for peptide reconstitution because the ionic strength of sodium and chloride ions destabilizes hydrogen bonding in peptides with charged amino acid residues. This causes aggregation and precipitation in compounds like Sermorelin, CJC 1295, and Ipamorelin, reducing potency by 30–50% within the first week. Additionally, saline lacks bacteriostatic preservatives, so it carries the same multi-dose contamination risk as sterile water. BAC water for mixing peptides is the only solvent that maintains both peptide stability and microbiological safety across a 28-day protocol.
Can I freeze reconstituted peptides to extend their shelf life?
▼
Yes, but only if you snap-freeze at −80°C in single-use aliquots to prevent ice crystal formation that ruptures peptide structure. Slow freezing in a standard −20°C freezer creates large ice crystals that mechanically shear peptide bonds, reducing potency by 10–15% per freeze-thaw cycle. For long-term storage, aliquot the reconstituted peptide into individual insulin syringes and freeze them separately — this eliminates repeated freeze-thaw cycles and maintains potency for 3–6 months. Never refreeze a thawed peptide dose, as each cycle compounds the degradation effect.
What does cloudiness in a reconstituted peptide vial indicate?
▼
Cloudiness indicates either peptide aggregation or bacterial contamination, both of which render the solution unsafe and ineffective for research use. Aggregation occurs when peptide chains clump together due to improper reconstitution technique — such as shaking instead of swirling, using cold water on frozen peptides, or prolonged storage beyond stability limits. Bacterial contamination appears as cloudiness, floating particles, or color change and signals complete loss of sterility. A properly reconstituted peptide should be crystal clear with no visible particulates — any deviation from this means the vial is compromised and should be discarded immediately.
Where can I source pharmaceutical-grade BAC water for peptide research?
▼
Pharmaceutical-grade bacteriostatic water must be sourced from USP-certified suppliers that provide certificates of analysis verifying 0.9% benzyl alcohol concentration and sterility testing. Real Peptides offers USP-grade Bacteriostatic Water specifically formulated for research peptide reconstitution, manufactured under strict quality controls to ensure consistent benzyl alcohol content and absence of particulates. Avoid non-certified sources or veterinary-grade bacteriostatic water, which may contain contaminants or incorrect preservative concentrations that compromise peptide stability and introduce safety risks.